You live in a time when your DNA is no longer just a mysterious code locked away in your cells. Bit by bit, scientists are turning that code into something you and your doctor can actually use to prevent disease, choose the right treatments, and even design healthier lives. What used to sound like distant science fiction is quietly slipping into clinic guidelines, lab reports, and routine checkups.
As you look at these discoveries, you’ll notice a pattern: the human genome keeps refusing to be simple. The more closely you examine it, the more layers appear – genes that are really switches, “junk” that isn’t junk at all, and risk scores that combine thousands of tiny nudges from your DNA. You do not need to be a geneticist to appreciate what this means. You just need to understand how these breakthroughs are reshaping the decisions you make about your health.
1. Your Genome Is a Health Map, Not a Life Sentence
You might have grown up hearing that genes are destiny, as if a single “bad” gene locks you into a certain future. The modern view of the human genome is almost the opposite: your DNA is more like a long, detailed health map that shows risks, routes, and detours, but not fixed outcomes. For many common conditions – heart disease, type 2 diabetes, some cancers – your risk is spread across thousands of small genetic variants that each nudge the odds just a little.
That means your genome matters most when you read it together with your environment and lifestyle, not in isolation. A high genetic risk for heart disease can be partly offset if you start statins earlier, exercise more, and pay closer attention to blood pressure and cholesterol. On the other hand, a seemingly “average” genetic risk can still turn into serious disease if you layer smoking, obesity, and chronic stress on top. You are not stuck with the map you were given, but you are responsible for how you use it.
2. Polygenic Risk Scores: Thousands of DNA Clues Rolled into One Number

Imagine if your doctor could look at a single number that summarizes how your entire genome affects your risk for a disease like coronary artery disease or breast cancer. That is what polygenic risk scores are starting to do. Instead of focusing on one rare mutation, they add up the impact of many common variants scattered across your DNA, each of which adds a tiny bit of risk or protection. With modern statistical methods and giant biobanks, that “tiny” information suddenly becomes powerful.
You are already seeing these scores move from research papers into real-world medicine. Cardiology guidelines are beginning to acknowledge them for coronary artery disease risk, and large networks have returned genome-informed risk assessments – including polygenic scores – to tens of thousands of patients. At the same time, insurers and some professional bodies still call routine polygenic testing “not medically necessary” because evidence and equity questions remain. For you, the takeaway is simple: polygenic scores are promising tools, but they are best treated as one more piece of the puzzle, not an absolute verdict.
3. The So-Called “Junk” DNA Is Driving Brain and Immune Health
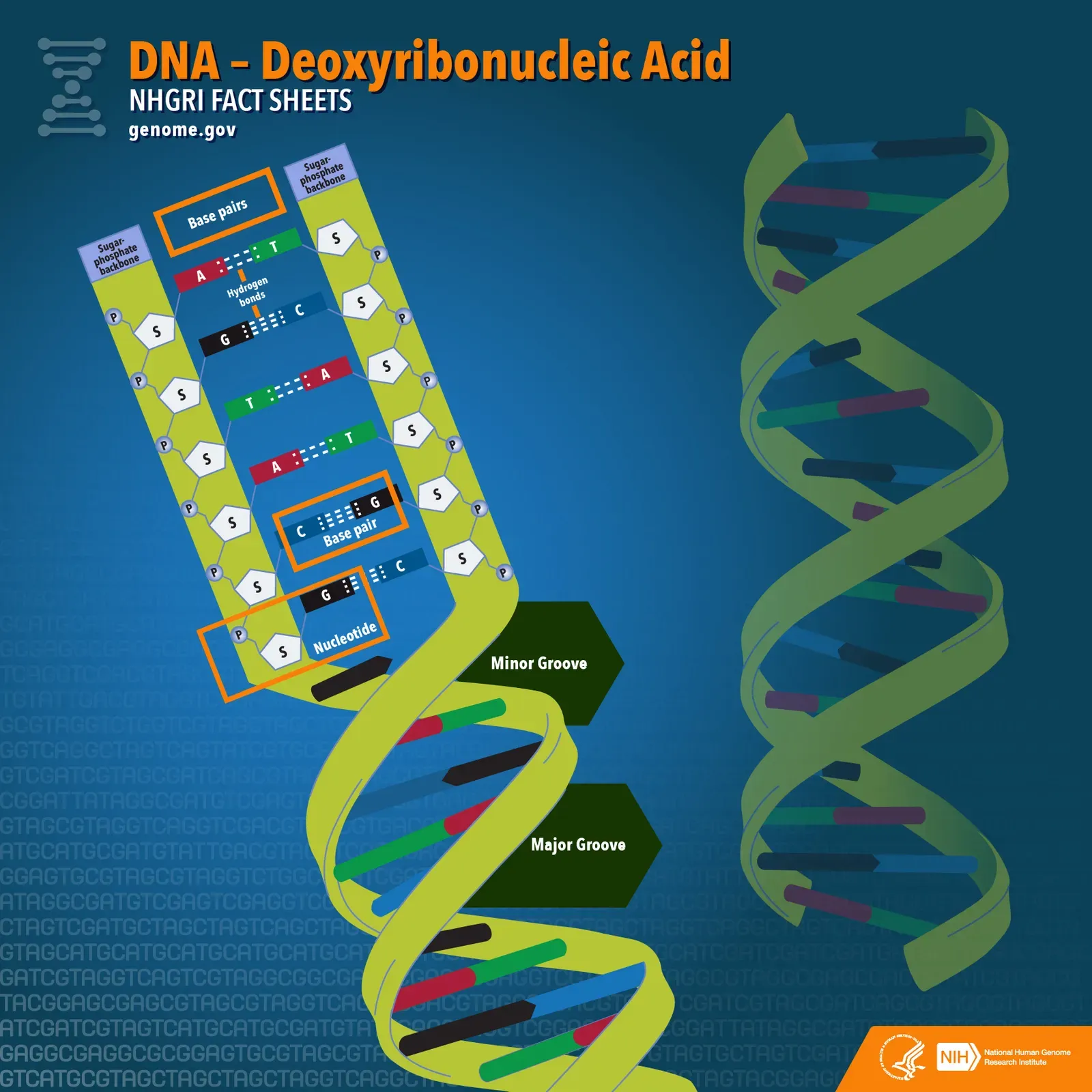
You were probably taught that only a tiny slice of your genome actually “does” anything, and the rest is useless junk. That story is rapidly falling apart. Roughly about one to two percent of your DNA codes for proteins, but the other ninety-plus percent – the vast noncoding regions – turn out to contain millions of regulatory switches. These switches help decide when, where, and how strongly your genes turn on, especially in your brain and immune system.
Recent work has linked specific noncoding “switches” to conditions like Alzheimer’s disease and other brain disorders, by testing how thousands of these elements behave in human brain cells. Similar efforts in blood stem cells and other tissues are revealing regulatory variants that change chromatin, the way DNA is packaged and read. For you, this means that rare coding mutations are only one part of the story; seemingly “silent” regions of your genome can shape how your brain ages, how you respond to infection, and how your body reacts to medications.
4. Ancient Viral DNA in You Is Helping Control Your Genes
You carry ancient viruses in your genome – literally. Over millions of years, retroviruses infected your ancestors and left pieces of their genetic material behind, stitched into human DNA. For a long time, those bits were dismissed as useless fossils. Now, you are seeing a very different picture: some of those viral sequences act like powerful regulatory elements that help control gene activity, particularly in early development.
In practical terms, this means that an infection that happened in a distant ancestor can still influence how your genes behave today. Certain viral-derived elements act like enhancers, turning nearby genes on or off at crucial moments, including in the developing embryo and in the brain. Understanding which of these viral fragments are active can help researchers pinpoint new drug targets and explain why some people seem unusually vulnerable – or surprisingly resilient – to autoimmune disease, neurodevelopmental conditions, and cancer. Your genome is not just human; it is a layered history of infections that your body has learned to repurpose.
5. Noncoding RNAs: The Invisible Conductors of Cellular Behavior
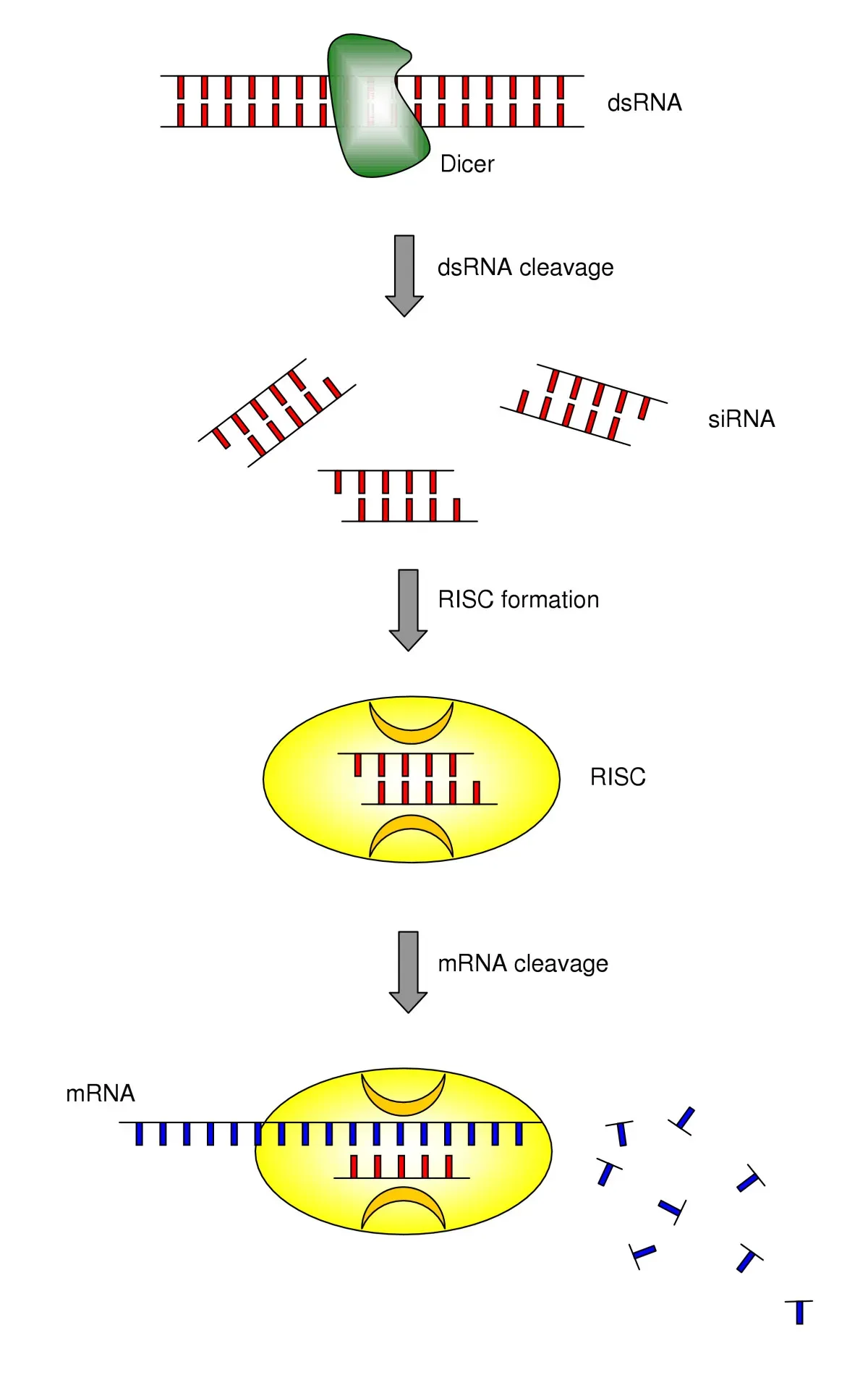
You might think of RNA simply as the messenger that carries DNA instructions to make proteins, but that is only part of the story. Your genome devotes a huge amount of effort to making RNAs that never become proteins at all. These long noncoding RNAs, microRNAs, and other exotic RNA species act more like conductors than musicians, coordinating which genes sing loudly and which stay quiet. Some safeguard your genome when DNA is damaged; others help your muscles repair themselves or steer stem cells toward a specific fate.
Tools that target RNA directly – using CRISPR systems designed to cut or block RNA molecules – have uncovered hundreds of noncoding RNAs that your cells rely on. Researchers have already found long noncoding RNAs that predict how brain tumors will behave, and others that control red blood cell development or maintain genomic stability. For you, this opens the door to therapies that do not touch your DNA at all but instead tweak the RNA layer, turning pathological programs down or restoring beneficial ones. It is like editing the conductor’s score without rebuilding the instruments.
6. Single-Cell Genomics Lets You Watch Disease Begin Cell by Cell
When you hear about “sequencing the genome,” you might picture one big readout for your whole body. Single-cell genomics flips that idea on its head by measuring DNA or RNA in individual cells, one by one. Techniques that can capture both DNA variants and RNA expression together in the same cell allow researchers to link a specific genetic change to its functional effect in real time. You are no longer looking at an average across millions of cells; you are watching distinct cell types misbehave as disease begins.
This matters because many conditions – cancer, autoimmune disease, neurodegeneration – start with rare cells that go off course long before symptoms appear. With single-cell tools, scientists can see which immune cells are misprogrammed in lupus, which neurons are vulnerable in Parkinson’s, or which tumor cells are likely to resist chemotherapy. That kind of detail can eventually help your doctor pick a drug aimed at the exact cell population driving your disease, rather than blasting everything and hoping. It moves you closer to treatments that are tuned to the fine-grained realities inside your tissues.
7. Long-Range Genome Models Reveal Distant DNA Conversations
Your genome is not a tidy list of genes lined up like books on a shelf. It is more like a tangled set of phone lines, where distant regions loop around to talk to each other. For years, most computational models could only “see” very short stretches of DNA at once, missing these long-range interactions. New AI architectures and clever algorithms now let you model hundreds of thousands to millions of bases of DNA in one shot, capturing how widely separated segments work together to control gene activity.
What does that do for you? It helps predict how a single letter change in a far-away regulatory region might ripple through your genome to affect cancer risk or drug response. It lets researchers simulate the effects of structural changes – deletions, inversions, duplications – on gene networks instead of guessing. These models are also being linked to electronic health records and polygenic risk scores so that your DNA patterns can be understood in the context of your entire clinical history. In the long run, that could help flag high-risk patients earlier and suggest tailored prevention strategies.
8. Genome Editing Is Getting Bigger, Smarter, and Sometimes CRISPR-Free
When you think of genome editing, you probably think of CRISPR snipping DNA at a single spot. In just a few years, the field has leapfrogged that basic idea. Researchers have shown that it is possible to manipulate stretches of DNA spanning up to around one million bases, not just a few letters. Others are harnessing “jumping gene” enzymes that can insert or delete DNA without relying on CRISPR at all, offering new routes to rewrite the genome more gently and precisely.
For you, this means therapies can start to tackle diseases caused by large deletions or complex structural rearrangements, not only single-point mutations. It also means the toolbox is diversifying: base editors that change one letter without cutting, prime editors that rewrite short regions, and transposase-based systems that can drop in therapeutic cassettes. Of course, that power comes with serious ethical and safety questions – especially around germline editing and embryo selection – so responsible use lags behind raw capability. Still, the fact that such large-scale edits are even technically possible is reshaping how you imagine future cures.
9. AI and Smarter Sequencing Tools Are Cleaning Up the Genome’s “Noise”
If you have ever tried to read a blurry photo, you know how hard it is to trust what you are seeing. Traditional sequencing faces a similar problem: signals are noisy, and mapping them to the reference genome is tricky. New software and AI-based tools are dramatically improving this step, aligning raw electrical signals from nanopore sequencing more accurately and catching errors that older models missed. That may sound technical, but it touches everything downstream.
With cleaner data, your genetic test results become more reliable, rare variants are less likely to be misread, and subtle patterns – like epigenetic marks or small structural variants – are easier to detect. These advances feed back into better disease-gene discovery, more accurate polygenic scores, and more trustworthy diagnostics. If you ever get your genome or exome sequenced, there is a good chance that behind the scenes an algorithm has already triple-checked the raw data so that your doctor is not making decisions based on a glitch.
10. Genome-Informed Clinical Models Are Changing How Risk Is Calculated

In the past, your doctor calculated your risk of something like a heart attack using age, cholesterol, blood pressure, smoking status, and maybe family history. Genomic medicine is quietly weaving DNA into those formulas. Large networks have started integrating monogenic variants, polygenic risk scores, and family history into electronic health records, building “genome-informed” profiles that can be used alongside lab values and imaging. Some experimental models even treat genetic risk as just another data stream in large health AI systems.
As this approach matures, you might see your preventive care plan shift. A high genomic risk for cardiovascular disease could tip the scales toward earlier statin therapy, more aggressive blood pressure targets, or more frequent imaging. A cluster of cancer risk variants might prompt earlier or more sensitive screening. At the same time, professional guidelines and payers are still catching up, and not every proposed use is supported by strong evidence. For you, the smart move is to treat genomic risk as a way to refine – not replace – the classic risk factors you already know.
11. Equity, Ethics, and Representation Are Becoming Central Genomic Questions

You cannot talk honestly about genomic medicine without talking about who is included. Most of the big genome-wide association studies that underpin polygenic risk scores have historically focused on people of European ancestry. That means if you are from a different background, a lot of the risk predictions built so far may be less accurate for you. Researchers are now openly acknowledging this gap and working to build multi-ancestry scores and more diverse biobanks, but the imbalance is not yet fully fixed.
On top of that, you face thorny ethical choices. Some fertility clinics have started offering embryo selection based on polygenic scores, even while many experts urge caution about accuracy, consent, and the specter of modern eugenics. Insurers and health systems must decide how to use genetic risk without penalizing vulnerable groups or deepening disparities. As someone who may one day be offered genome-guided interventions, you benefit from staying skeptical, asking who the data came from, and demanding transparency about limitations. The genome may be a marvel of biology, but how you choose to use it is a social and moral decision.
Genomic discoveries are no longer abstract science; they are creeping into guidelines, electronic health records, and clinical conversations that directly affect you. The most amazing part is not that your genome can now be read, but that it can help shape practical choices about screening, prevention, and treatment – without ever fully dictating your fate. As the map grows more detailed, the real question becomes: how will you, your doctors, and your society decide to follow it?




