Have you ever wondered how scientists discovered that atoms aren’t just solid little balls? Or how we figured out that life could emerge from nonliving chemicals? History is packed with moments where researchers took wild chances, pushed boundaries, and came up with results that seemed almost impossible.
Some of these experiments were so revolutionary that they completely overturned everything we thought we knew about reality. From gold foil that revealed secrets hiding inside atoms to flasks of chemicals sparked into something resembling the origins of life itself, these discoveries didn’t just make headlines. They rewrote the rules of science and changed our understanding of existence. Let’s dig into five experiments that were so unexpected, so bold, they transformed everything.
Rutherford’s Gold Foil Experiment: Finding the Heart of Matter
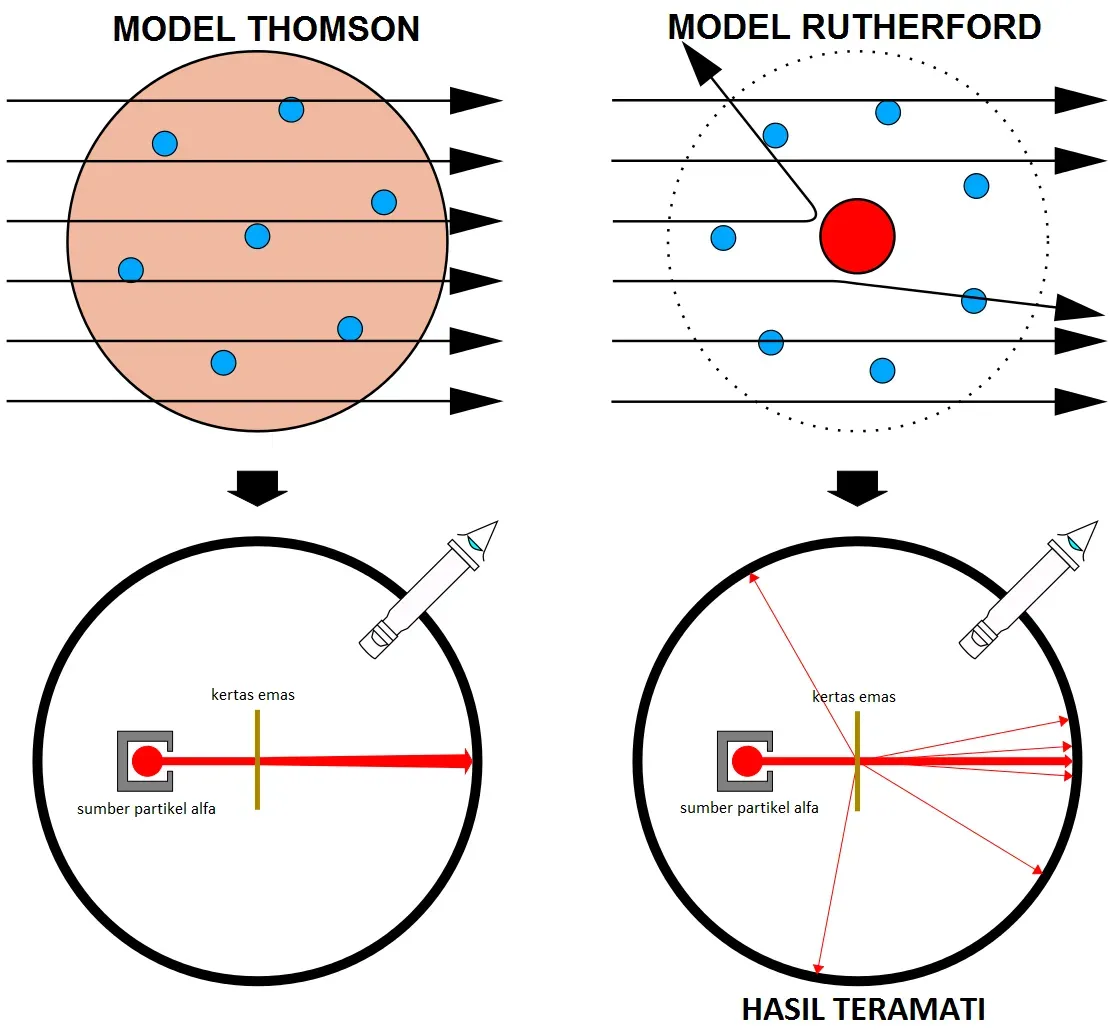
The Rutherford scattering experiments were a landmark series where scientists learned that every atom has a nucleus where all of its positive charge and most of its mass is concentrated, performed between 1906 and 1913 by Hans Geiger and Ernest Marsden under Ernest Rutherford’s direction at the University of Manchester. The experiment involved firing alpha particles from a radioactive source at a thin gold foil, and any scattered particles would hit a screen coated with zinc sulfide, which scintillates when hit with charged particles. What they expected was simple. What they got was mind blowing.
Marsden saw lots of tiny, fleeting flashes of yellowish light, on average more than one blip per second. As Rutherford was fond of saying, it was as if you fired a shell at tissue paper and it came back and hit you, with about one in every few thousand alpha particles scattered at an angle greater than 90 degrees. This didn’t fit the prevailing model of the atom at all. Rutherford suggested in 1911 that alpha particles were being scattered by a large amount of positive charge concentrated in a very small space at the atom’s center, proposing that electrons must be orbiting around this central core like planets around the sun, and he calculated the nucleus to be only about one hundred thousandth the size of the atom. The atom was mostly empty space, a fact that still amazes people today.
The Miller Urey Experiment: Cooking Up Life in a Flask
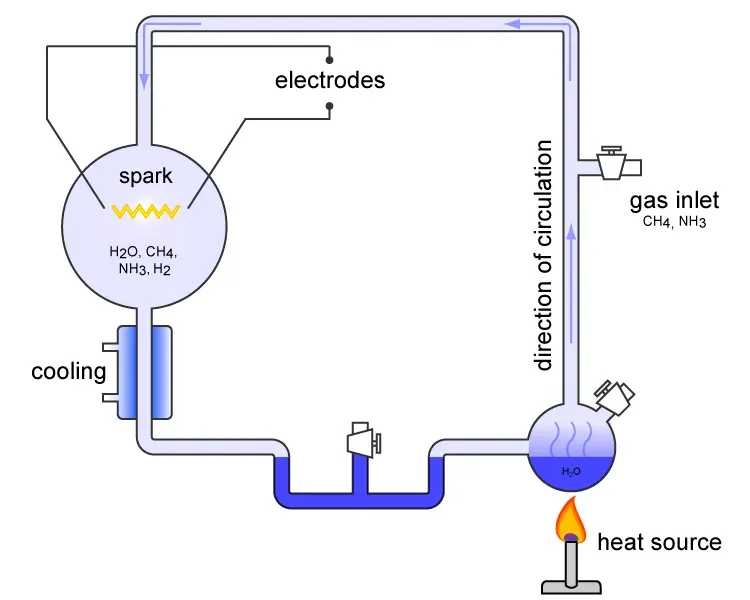
Let’s be real, the question of how life began has haunted humanity forever. The Miller Urey experiment was carried out in 1952 and simulated the conditions thought at the time to be present in the atmosphere of the early, prebiotic Earth. Stanley Miller, then a graduate student at the University of Chicago, approached Harold Urey about doing an experiment to evaluate whether organic compounds important for the origin of life may have been formed abiologically on the early Earth, mimicking lightning by the action of an electric discharge on a mixture of gases representing the early atmosphere.
A red and yellow coloured solution started to collect after running the experiment for a few days and became a broth of red and brown by the end, and they discovered several types of simple organic molecules in the samples, including amino acids, some of which were relevant as the building blocks of proteins present in all living organisms. Here’s the thing. The experiment showed that amino acids, important components of proteins critical to life on Earth, could have arisen from inorganic compounds during Earth’s prebiotic phase, and it demonstrated that the speculation that life could have originated through chemical reactions among nonliving materials is possible and that this hypothesis could be tested scientifically. Though our understanding of Earth’s early atmosphere has evolved since then, the experiment opened an entirely new field of research into how life could emerge from chemistry.
Pasteur’s Swan Neck Flask: Death to Spontaneous Generation

Back in the 1800s, people thought food spoiled and diseases were caused by bad air or life spontaneously generating, and Louis Pasteur rejected the idea that mice could be randomly created from rotting wheat and old cloth over a few weeks. Sounds ridiculous now, right? Yet this belief persisted for centuries. After noticing that his own vats of beer were turning sour, Pasteur started analysing them only to discover they were swarming with bacteria, which convinced him that the spoiling of his brew was caused by these tiny microorganisms.
He designed a simple experiment to prove his revolutionary germ theory, disproving the idea that cells could come from nothing, and his work in the food and medicine industry was so crucial that we even named a process after him, pasteurisation, the process of heat treating something for a short time and cooling it down quickly to make it safe from bacteria. The experiment was elegant in its simplicity. Pasteur showed that life doesn’t just pop into existence. It comes from other life. This insight saved countless lives by transforming our approach to food safety and medicine, proving that sometimes the simplest experiments deliver the biggest punches.
Mendel’s Pea Plants: Cracking the Code of Inheritance
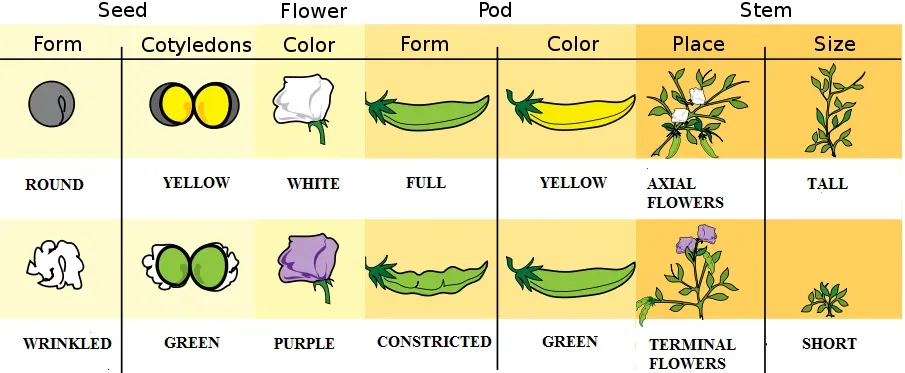
The answer to how we inherit our genes from our parents was actually discovered not by studying humans but peas, as Gregor Johann Mendel, an Augustinian friar, crossbred peas with differing characteristics to evaluate how different features were inherited in their offspring, with his work focusing on pea plants and their seven observable traits including the shapes of the pods and seeds, plant height, flower position, and seed, pod and flower colour. Working quietly in a monastery garden, Mendel conducted thousands of careful crosses and kept meticulous records. Most people thought his work was boring at the time.
The profound mystery behind the inheritance of physical traits began to unravel a century and a half ago thanks to Mendel, and ensconced at a monastery in Brno, the shy Gregor quickly began spending time in the garden. Fuchsias grabbed his attention, their daintiness hinting at an underlying grand design, and he had been crossing different varieties, trying to get new colours or combinations of colours, and he got repeatable results that suggested some law of heredity at work. His discoveries became the foundation of modern genetics. Honestly, it’s wild to think that the secrets of DNA, genetic diseases, and inheritance all trace back to a quiet monk growing peas in a garden, carefully counting wrinkled versus smooth seeds.
Fermi’s Chicago Pile: Unleashing the Atomic Age

On December 2, 1942, as World War II raged, a small group of scientists gathered beneath the football stands of the University of Chicago for a physics experiment that would literally change the world, having constructed in utmost secrecy a pile of uranium and graphite that would soon become the very first nuclear reactor, as Enrico Fermi and the scientists hoped to split uranium atoms and create the world’s first self sustained, controlled chain reaction. The stakes couldn’t have been higher. They were essentially playing with the fundamental forces of the universe in the middle of a major city.
The pile was 6 tons of uranium metal, 50 tons of uranium oxide, and 400 tons of graphite bricks laid in an arrangement painstakingly calculated by some of the greatest scientific minds in the country, and at 3:25 p.m., Fermi gave the final signal, the counters ticked, and the pile achieved criticality, marking that the atomic age had begun. Seventy six years later, the implications of that physics experiment still reverberate around the world, shaping the outcome of World War II and the following half century of international foreign policy, and it has continued to shape the way the world views and uses energy, medicine, and scientific research. This experiment gave us nuclear power, medical treatments using radiation, and unfortunately, nuclear weapons. The weight of that moment under those football stands continues to echo through every debate about energy and weapons today.
Conclusion: When Curiosity Changes Everything

These five experiments share something remarkable. They all began with simple questions that seemed almost childish in their directness. What’s inside an atom? Where did life come from? Can life appear from nowhere? How do children resemble their parents? Can we control the power of the atom? The scientists who asked these questions didn’t have fancy equipment or unlimited budgets initially. They had curiosity, determination, and the courage to challenge what everyone else believed.
Each of these discoveries fundamentally altered humanity’s trajectory. We went from thinking atoms were solid lumps to understanding they’re mostly empty space with a dense nucleus. We shifted from believing life magically appeared to understanding chemical processes could create its building blocks. We stopped thinking disease arose from bad air and started fighting germs. We decoded the rules of inheritance that govern all life. We learned to harness nuclear power for better or worse.
What’s truly mind blowing is that we’re still building on these foundations today in 2026. Modern genetics, nuclear medicine, food safety protocols, our search for life on other planets – all of it traces back to these wild experiments that seemed impossible at the time. It makes you wonder what seemingly crazy experiments happening right now in labs around the world will be in history books a century from now. What do you think the next world changing discovery will be?

Hi, I’m Andrew, and I come from India. Experienced content specialist with a passion for writing. My forte includes health and wellness, Travel, Animals, and Nature. A nature nomad, I am obsessed with mountains and love high-altitude trekking. I have been on several Himalayan treks in India including the Everest Base Camp in Nepal, a profound experience.




