If you could rewind Earth’s history like an old tape, you’d eventually hit a strange, almost alien chapter: a world without animals, plants, or even proper cells. Somehow, from a planet of rock, water, and violent storms, life learned how to spark, copy itself, and evolve into everything we see today. The wild part is that scientists are finally starting to piece together how that impossible-sounding leap might have happened.
Over just the past couple of decades, new experiments, space missions, and deep-ocean expeditions have completely shaken up old ideas about how life began. The story that’s emerging is far more dramatic, messy, and creative than the simple “primordial soup” picture many of us learned in school. It’s a tale of lightning, meteorites, volcanoes, microscopic bubbles, and molecules that behave almost like characters in a thriller. Let’s walk through ten of the most surprising discoveries changing what we thought we knew about our own origins.
1. The “Primordial Soup” Experiment That Started It All Still Holds Up
One of the most famous experiments in science, done in the 1950s, showed that simple gases and sparks can create building blocks of life. Researchers mixed gases thought to resemble early Earth’s atmosphere, zapped them with electrical discharges like artificial lightning, and found that amino acids had formed. For decades, people wondered if that classic experiment was too simple or unrealistic compared to the chaotic early planet.
Here’s the twist: more recent work has shown that versions of this “primordial soup” process can still produce a surprising variety of organic molecules under different conditions. Scientists have repeated and updated the original experiment with more realistic gas mixtures, mineral surfaces, and energy sources like ultraviolet light. They keep finding that basic ingredients of life can emerge fairly easily when you have energy, simple chemicals, and time. It doesn’t prove exactly how life began, but it tells us that nature can cook up life’s starter kit without much help.
2. Deep-Sea Hydrothermal Vents Look Like Natural Life-Forging Laboratories

Imagine a pitch-black ocean floor, cut by cracks in the crust where superheated water gushes out like underwater geysers. These hydrothermal vents, discovered in the late twentieth century, turned out to be bustling with strange life that doesn’t rely on sunlight at all. Instead, microbes there feed on chemical energy from the Earth’s interior. That alone was shocking enough, but it also gave researchers a new idea for where life might have begun.
Some of these vents, called alkaline hydrothermal vents, form porous mineral structures full of tiny compartments. Inside them, natural chemical gradients look a bit like the voltage across modern cells. That made scientists wonder if these ancient vents acted like living batteries, helping early molecules gain energy and self-organize. In lab simulations, similar mineral pores can concentrate organic molecules and drive simple reactions, hinting that the first life might have been born in the dark, not in a warm surface pond.
3. Life’s Building Blocks Rain Down from Space More Often Than You’d Think

It sounds like science fiction, but some of the ingredients for life literally fall from the sky. Analyses of certain meteorites and cometary material have revealed amino acids, sugars, and other organic compounds. When spacecraft and telescopes studied comets and asteroids more closely, they kept finding more and more complex carbon-based chemistry hiding in the cold of space.
Even dust grains floating in interstellar clouds show signs of organic molecules forming on their surfaces. That doesn’t mean life itself arrived fully formed from space, but it does mean the universe is much more chemically creative than we once believed. Early Earth was constantly bombarded by rocks and ice loaded with these compounds, like a delivery service dropping off raw ingredients. Instead of starting from scratch, our planet may have received a massive head start from the cosmos.
4. RNA Might Have Been the First Molecule to Behave Like Both Gene and Tool
Modern life uses DNA to store information and proteins to do the heavy lifting, but that raises a chicken-and-egg problem: which came first? One influential idea is that RNA, a cousin of DNA, may have played both roles in a simpler, earlier world. In the late twentieth century, researchers found that some RNA molecules can fold into shapes that let them act like tiny tools or catalysts, speeding up chemical reactions.
This discovery made the so-called “RNA world” hypothesis suddenly feel more realistic. Scientists have since shown that RNA can, under the right conditions, help copy itself and perform basic tasks without proteins. It still leaves a lot of unanswered questions, like how the first long RNA strands formed in the wild without help. But it suggests that life might have started with a single versatile molecule that gradually handed off jobs to more specialized partners as complexity grew.
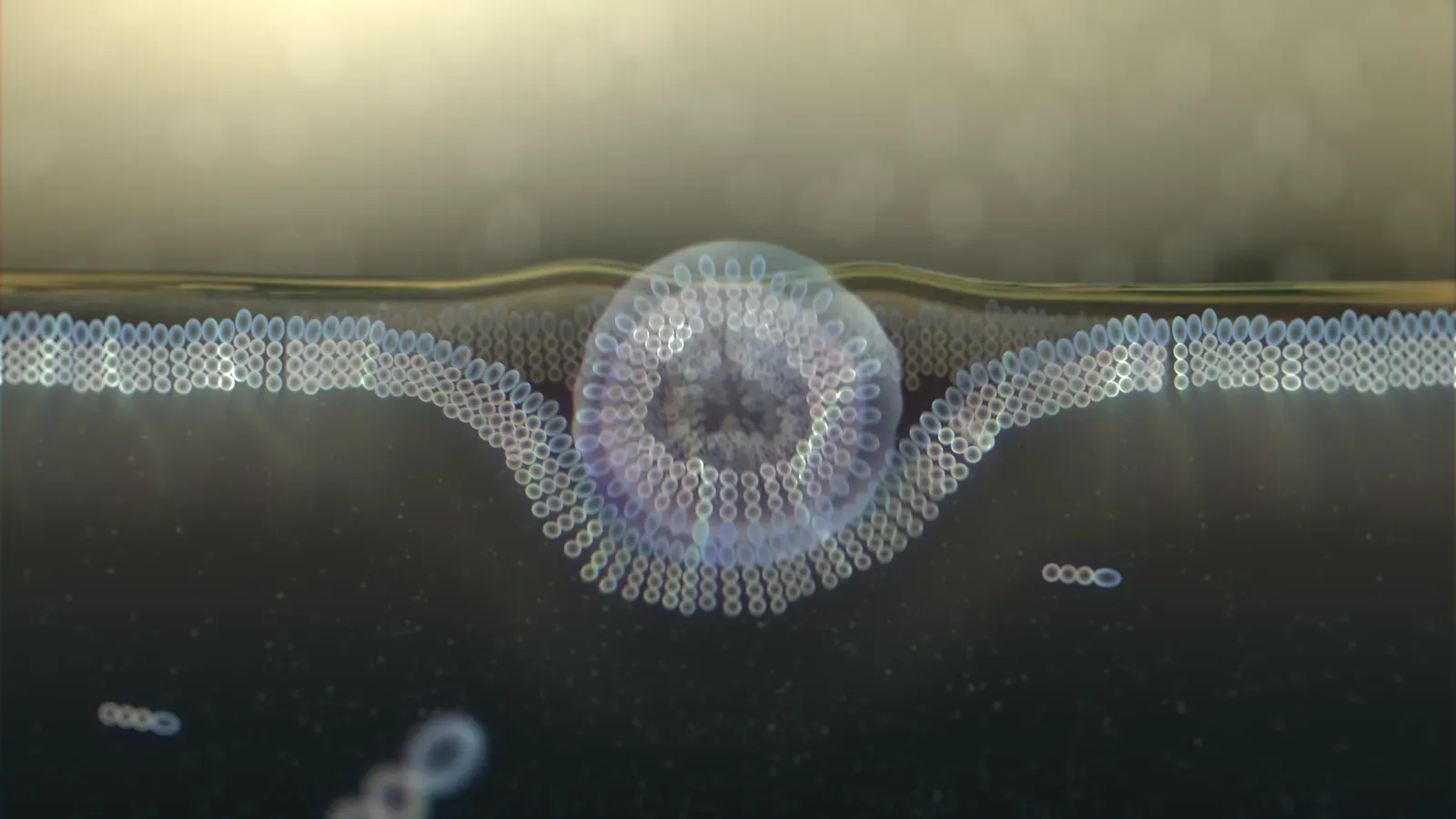
5. Simple Fatty Bubbles Can Form Cell-Like Compartments on Their Own
Life as we know it depends on cells: tiny, enclosed spaces where chemistry can happen in a controlled way. For a long time, it wasn’t clear how the very first cells could have formed without the complex machinery we see today. Then research on simple fatty molecules changed the picture. It turns out that under the right conditions, certain fatty acids spontaneously arrange themselves into small spheres, called vesicles, in water.
These bubbles look eerily like primitive cell membranes. In experiments, they can grow, divide, and sometimes even trap RNA or other molecules inside by accident. When you see that, it becomes easier to imagine early Earth dotted with countless microscopic bubbles, each acting like a little test tube. In some of them, lucky combinations of molecules could have started to cooperate, setting the stage for the first truly living cells to emerge from these fragile, soap-bubble-like shells.
6. Minerals May Have Been the Unsung Matchmakers of Early Chemistry

Rocks might not seem like they have much to do with life, but they can be surprisingly helpful partners in chemistry. Experiments have shown that certain minerals can help link small molecules together into longer chains, which is crucial for making things like RNA or simple proteins. Surfaces of minerals can hold onto molecules, bring them close together, and even guide their arrangement, a bit like a workbench helping a builder line up pieces.
Some iron and clay minerals appear particularly good at catalyzing these early steps. In simulated early Earth conditions, they can concentrate organic molecules from dilute solutions and push reactions in directions that might otherwise be unlikely. This makes the idea of life emerging on a bare rock planet less mysterious: the crust itself may have acted as a silent collaborator. When you picture early Earth now, it’s not just random soup; it’s a planet covered in reactive surfaces constantly nudging chemistry forward.
7. Ancient Rocks Carry Chemical Whispers of Early Microbes

We obviously don’t have fossils of the very first cells, but we do have clues buried in some of the oldest rocks on the planet. Geologists have found ancient formations more than three billion years old that show layered structures often linked with microbial communities. In some cases, the ratios of certain forms of carbon locked inside these rocks look like the fingerprints of biological activity rather than pure geology.
These signals are subtle, and scientists debate exactly how to interpret some of them. Still, taken together, they suggest that microbial life appeared on Earth surprisingly quickly once the planet cooled enough to be habitable. That speed is startling: it hints that, under the right conditions, life might not be incredibly rare after all. When I first learned that, it made the universe feel both more crowded and more mysterious at the same time.
8. Extreme Microbes Prove Life Thrives Where We Once Thought It Impossible
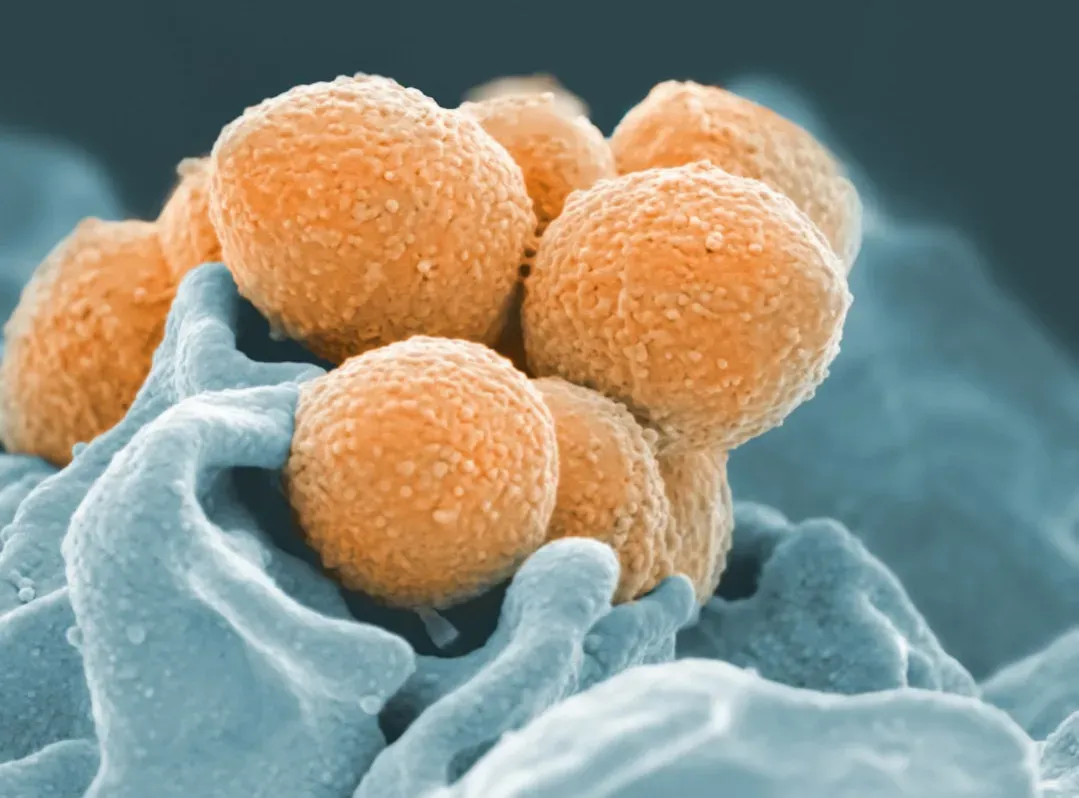
One of the biggest shifts in thinking came from discovering extremophiles: organisms that live in conditions once thought completely hostile to life. Microbes have been found in boiling hot springs, acidic lakes, deep underground rocks, and even inside Antarctic ice. Some survive crushing pressures at the bottom of the ocean, or near-freezing brines that would destroy most familiar cells.
These hardy life forms don’t just stretch our imagination; they reshape theories about how and where life could begin. If modern organisms can adapt to such extremes, then early life might have started in places we used to rule out, like super-salty pools or scorching volcanic shorelines. It also raises the possibility that other worlds with harsh environments, such as icy moons or Mars, might be more promising for life than we once dared to hope. The boundary between “habitable” and “impossible” looks a lot fuzzier now.
9. The Early Earth Was More Violent and Yet Weirdly Hospitable Than We Imagined

For a long time, scientists thought that early Earth went through a prolonged period of intense bombardment by giant space rocks that would have sterilized the surface repeatedly. That scenario made it tricky to explain how life could get started and survive. More recent dating of ancient minerals and lunar samples suggests that the late heavy bombardment may have been shorter and less uniform than once believed.
At the same time, evidence from old zircons, tiny crystals that act like time capsules, indicates that liquid water and even oceans existed quite early in Earth’s history. That combination of violent impacts and stable water is strange but encouraging for life’s origin. It paints a picture of a world that was dangerous but also full of sheltered niches, like cracks, pools, and deep-sea environments where fragile chemistry could take hold and gradually toughen up. Life may have threaded its way through chaos rather than waiting for a perfectly calm moment.
10. The Line Between Chemistry and Life Is Getting Blurry in the Lab
Perhaps the most mind-bending discovery is that researchers are now building systems in the lab that start to resemble something in between non-living chemistry and true life. Scientists have created simple chemical networks that can copy patterns, adapt to changing conditions, or even compete for resources. These are not alive in the way a cell is, but they hint at how lifelike behavior can gradually emerge from ordinary reactions.
There are also experiments where droplets of chemicals show primitive forms of movement, growth, or division, driven only by simple physics and chemistry. Watching these systems, it becomes easier to picture how matter can slowly edge toward life without a sharp dividing line. It’s less like flipping a switch and more like walking up a long, winding ramp from chaos to complexity. Seeing that transition start to appear in glass flasks and microfluidic chips makes the origin of life feel less like a miracle and more like a difficult, but understandable, puzzle.
A Story Still Being Written in Rock, Water, and Starlight

When you put all these discoveries together, the origin of life stops looking like a single lucky lightning strike and starts to resemble a whole planet experimenting with chemistry for millions of years. Deep-sea vents, volcanic pools, mineral surfaces, fatty bubbles, and cosmic rain all contribute different pieces to the same sprawling story. No single theory has won outright, and it’s very possible that life’s beginnings involved a messy mix of several pathways.
What’s most striking is how often nature seems to nudge matter toward complexity whenever the conditions are right. Molecules self-assemble, reactions organize, structures emerge, and patterns persist, long before anything we’d clearly call alive appears. The more closely we look, the more it feels as if life is not an abrupt exception to the universe but one of its natural outcomes. Knowing that, it’s hard not to wonder what other worlds might be quietly running their own slow experiments in turning chemistry into something that can look back and ask how it all began.