At the start of this century, many scientists quietly believed we already understood the broad strokes of how the human body works. Two decades later, that confidence looks almost quaint. From hidden organs and microscopic ecosystems to immune tricks and brain rewiring, research since the year 2000 has revealed that our bodies are stranger, more dynamic, and more interconnected than most textbooks ever suggested. These discoveries are not just scientific curiosities; they’re reshaping how we diagnose disease, design drugs, and even think about what it means to be human. Here are ten breakthroughs that show just how wildly the story of the human body has changed in only a few short decades.
1. The Microbiome Revolution: You Are Not Just You
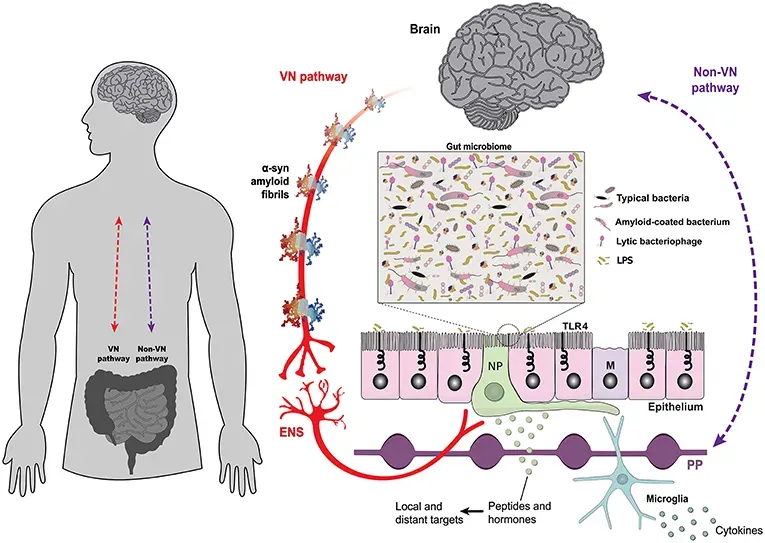
Imagine discovering that for every human cell in your body, there are roughly about as many microbial cells along for the ride. That realization, driven by large-scale microbiome studies launched in the early 2000s, overturned the old idea that microbes were mostly invaders to be killed or kept out. Instead, scientists found bustling communities of bacteria, viruses, and fungi living in your gut, on your skin, and even in your mouth, shaping everything from digestion to immunity and mental health. These microbial partners help break down complex fibers, produce vitamins, train your immune system, and may influence mood and behavior through chemical signals to the brain.
What shocked researchers was just how individualized these microbial “fingerprints” are – no two people have identical microbiomes, not even identical twins. Patterns have emerged linking certain microbial mixes to obesity, inflammatory bowel disease, allergies, and possibly even depression and anxiety. Early trials of fecal microbiota transplantation, where stool from a healthy donor is transferred to a sick patient, have shown dramatic success in treating recurrent gut infections and are now being explored for other conditions. The human body, it turns out, is more like a coral reef than a single organism: a living system built from partnership, not isolation.
2. Hidden Anatomy: The Surprising “New” Structures Inside Us
It sounds like something from a Victorian adventure novel: modern scientists discovering “new” parts of the human body with 21st-century tools. Yet that’s exactly what has happened, as high-resolution imaging and careful re-examination of old specimens have revealed structures that were overlooked or misunderstood for years. One headline-grabbing example was the proposal in the late 2000s that the interstitium – a network of fluid-filled spaces in tissues – might be considered a kind of organ-like system involved in cushioning organs and transporting immune cells. Another example is the mesentery, a fold of tissue anchoring the intestines, which was reclassified by some researchers as a distinct organ based on structural and functional criteria.
Even more recently, detailed mapping has uncovered small salivary glands deep in the head that had previously evaded standard anatomical descriptions, highlighting how even familiar regions can hide surprises. These discoveries do not mean humans suddenly grew new body parts, of course; rather, they show how classification and attention shape what we see. When technology improves and someone is curious enough to look again, familiar landscapes can reveal hidden rooms and secret corridors. In a sense, these “new” structures remind us that anatomy is not a finished map pinned to a wall, but a living document still being edited.
3. Brain Plasticity: A Flexible Mind in a Changing Body
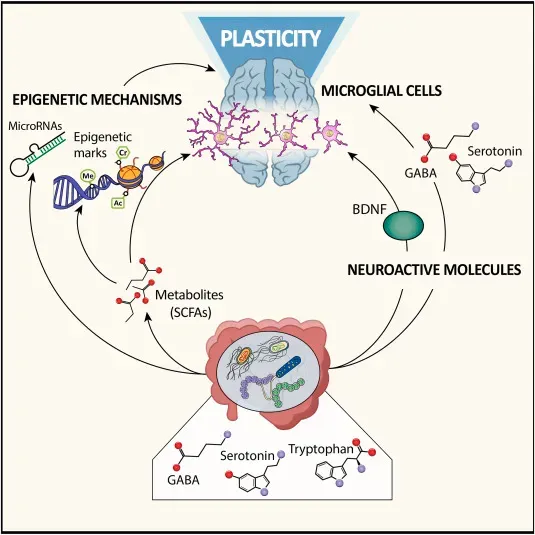
For much of the twentieth century, people were told that once the brain matured, it was essentially fixed, like concrete that had finally set. Research in the last two decades has shredded that idea. Neuroimaging and long-term clinical studies have shown that the adult brain is remarkably plastic, constantly rewiring connections in response to learning, injury, and experience. Stroke patients, for example, can sometimes regain lost abilities as other brain regions reorganize to take over damaged functions, especially when paired with targeted rehabilitation.
On a more everyday level, learning a new language, practicing an instrument, or even navigating a new city physically reshapes neural circuits, strengthening some pathways while pruning others. Studies have documented changes in the structure and function of brain regions involved in attention, memory, and emotion regulation after mindfulness training or cognitive-behavioral therapies. This plasticity has fueled a new wave of brain–computer interface research and neuroprosthetics, in which people with paralysis can learn to control robotic arms or computer cursors through patterns of brain activity. The modern view of the brain is less like a static machine and more like a muscle: built to change as long as life continues to present new challenges.
4. The Immune System’s Double Life: From Defense to Designer Tool
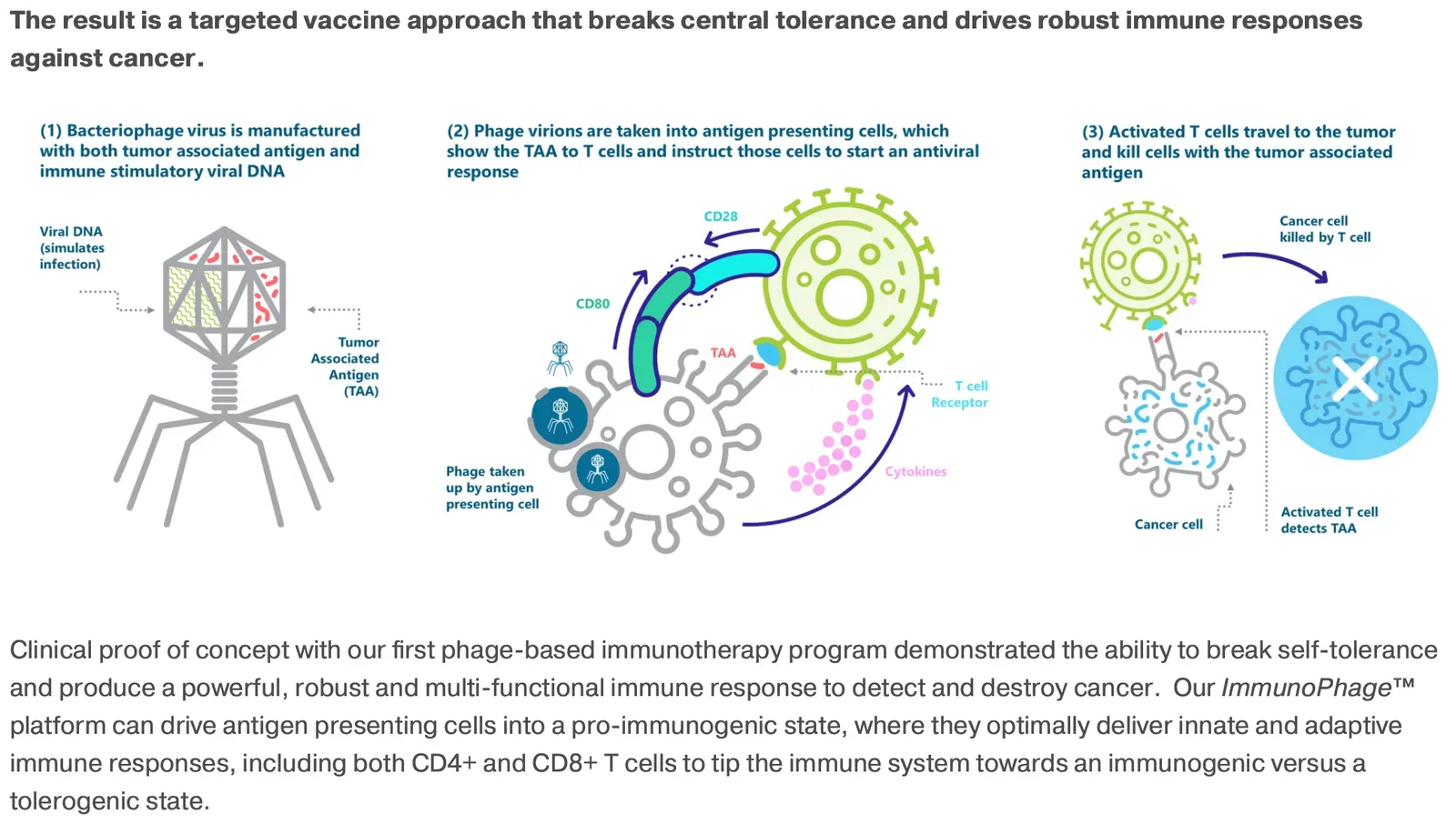
The human immune system was once described mainly as a defense force, a kind of microscopic army built to recognize and destroy invaders. Since the early 2000s, however, scientists have learned that those same sentinels can be reprogrammed and redirected, turning a natural defense into a powerful precision tool. One of the most striking examples is immunotherapy for cancer, including checkpoint inhibitors that release the “brakes” on immune cells and CAR-T therapies that genetically engineer a patient’s own T cells to hunt down tumors. These approaches have led to lasting remissions in some people with cancers that were previously considered almost hopeless.
At the same time, researchers have begun to unravel how immune cells talk to nearly every organ system, influencing metabolism, brain function, and even tissue repair. Misfires in this intricate signaling network are now linked to autoimmune diseases, long COVID, and chronic inflammation that can quietly damage organs over years. What we’ve learned this century is that immunity is less like a simple wall and more like a vast communications network, full of negotiation, memory, and feedback loops. By tapping into that network, medicine is moving toward treatments that work with the body’s own strategies instead of trying to overpower them from the outside.
5. Genomic Insights: From “Junk DNA” to Regulatory Goldmine
When the first draft of the human genome was announced in the early 2000s, many people focused on the relatively small slice that actually encodes proteins. Large swaths of the genome were dismissed as “junk,” evolutionary leftovers with little obvious function. Over the following years, that view has dramatically shifted. Projects that map gene activity and regulatory elements have revealed that non-coding regions are densely packed with switches, signals, and structural elements that control when and where genes are turned on. Far from being useless, these stretches of DNA are like a vast library of instructions, timing cues, and context.
Mutations in these regions are now known to contribute to a wide range of conditions, from heart disease and diabetes to neurodevelopmental disorders and some cancers. Researchers have also identified subtle variations in non-coding DNA that help explain why certain drugs work well for some people but not others, pushing forward the field of precision medicine. Gene editing tools such as CRISPR have given scientists a way to probe and sometimes correct specific sequences, transforming theoretical insights into experimental therapies. The genome, once seen as a mostly static blueprint, is now viewed as a dynamic script with intricate stage directions written between the lines.
6. Metabolism Reimagined: Brown Fat, Hunger Hormones, and Internal Clocks
If you grew up thinking of fat as a passive storage depot and metabolism as a simple calorie equation, modern research would feel like reading a plot twist. Early in the century, scientists confirmed that adults retain more brown adipose tissue, or “brown fat,” than once believed. Unlike white fat, brown fat burns energy to generate heat, and its activity is linked to cold exposure and metabolic health. At the same time, researchers have discovered a complex web of hormones – such as leptin and ghrelin – regulating appetite and satiety, signaling between fat tissue, the gut, the brain, and other organs.
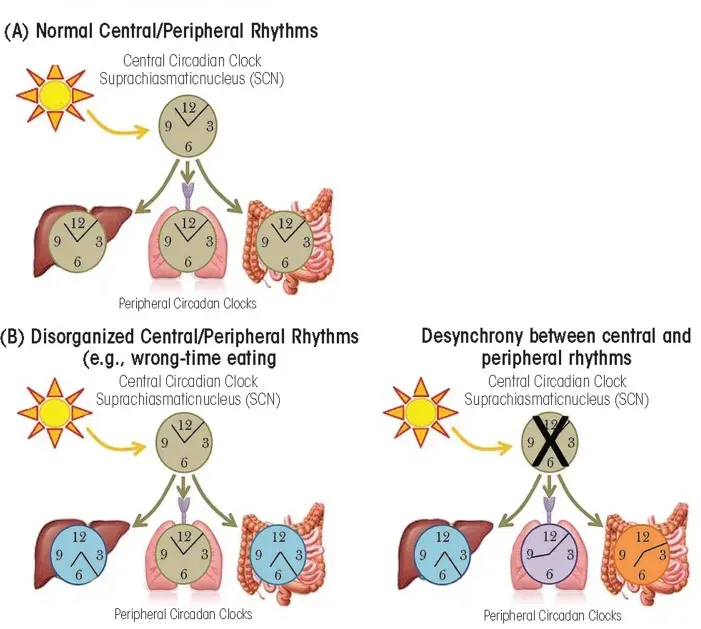
Circadian biology has layered yet another level of complexity, showing that nearly every cell in the body runs on internal clocks that govern when genes involved in digestion, hormone release, and energy use are most active. Disrupting these rhythms through chronic night-shift work or irregular sleep patterns has been associated with higher risks of obesity, diabetes, and cardiovascular disease. These findings help explain why two people with similar diets and activity levels can have very different metabolic outcomes. They also point toward interventions that go beyond calorie counting: aligning eating patterns with biological clocks, targeting brown fat activation, and designing drugs that modulate hunger hormones rather than simply suppressing appetite in blunt ways.
7. The Brain–Body Loop: How Emotions, Organs, and Nerves Intertwine
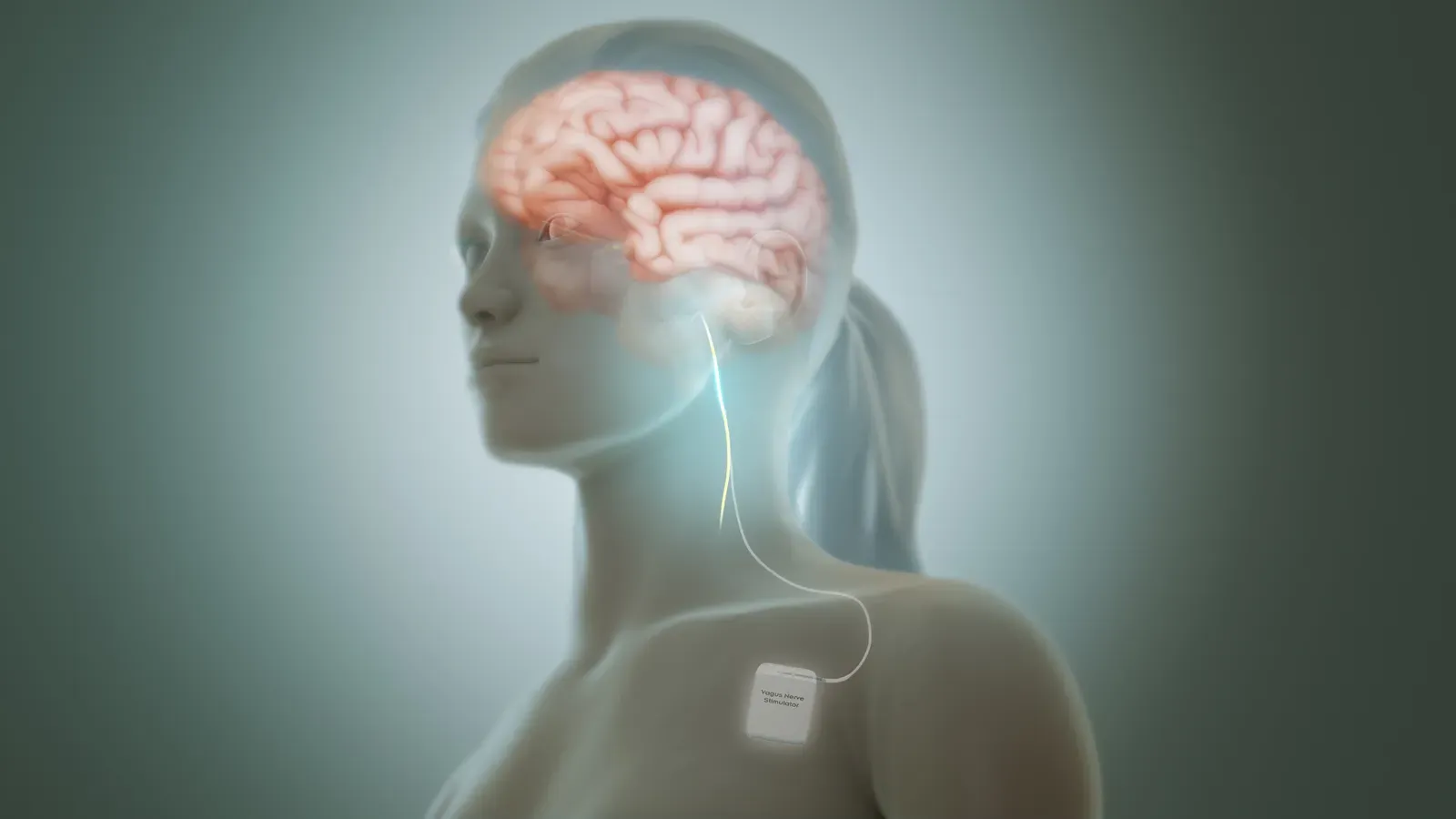
For much of modern history, people casually separated mind and body, as if thoughts floated somewhere above the neck and the rest of the organs just carried out mechanical tasks. This century’s research has taken a sledgehammer to that divide. Studies of the gut–brain axis have revealed dense networks of nerves and chemical messengers connecting the intestines to mood and cognition, suggesting that what happens in the digestive tract can influence anxiety, depression, and even decision-making. The vagus nerve, a major communication highway between organs and the brain, has emerged as a key player in transmitting signals about inflammation, heart rate, and internal stress.
Clinical trials using vagus nerve stimulation for conditions like epilepsy, depression, and inflammatory diseases have shown that altering these body–brain conversations can change both physical and psychological symptoms. Meanwhile, research on stress has documented how chronic emotional strain can reshape brain circuits, immune responses, and metabolic pathways, increasing vulnerability to disease. The picture that emerges is not of a mind housed in a body, but of a single integrated system in constant two-way dialogue. Feelings, it seems, are not just in your head; they echo in your gut, your heart, and your immune system as well.
8. Why These Discoveries Matter: Rethinking Health, Disease, and “Normal”
It might be tempting to treat each of these breakthroughs as a fun trivia fact – another surprising thing to drop into conversation at dinner. But together, they signal a deeper shift in how medicine understands the human body. Traditional approaches often treated diseases as isolated malfunctions in single organs: heart disease in the heart, depression in the brain, diabetes in the pancreas. This century’s discoveries emphasize networks instead: microbiomes interacting with immunity, genomes shaping responses to environment, and brain and body acting as a single complex system. That means the same condition can play out differently in different people, depending on their microbial communities, genetic variants, lifestyle, and histories.
Compared with older models that sought one-size-fits-all explanations and cures, the new picture is both more challenging and more hopeful. It explains why some standard treatments work brilliantly for some patients and barely at all for others, and it opens the door to more tailored strategies that take individual biology seriously. It also forces uncomfortable questions about what “normal” even means in a world where variation is the rule rather than the exception. In practical terms, these discoveries are slowly nudging healthcare away from purely reactive crisis management and toward prediction, prevention, and personalization. The stakes are not abstract; they show up in how long we live, how well we age, and how much suffering can be avoided along the way.
9. The Future Landscape: From Organs-on-Chips to Engineered Immunity
Standing in 2025, it already feels like we’re halfway into science fiction, and yet most researchers will tell you we’re still just scratching the surface. In labs around the world, organ-on-a-chip systems – tiny devices lined with human cells that mimic the behavior of lungs, kidneys, or guts – are being used to study disease and test drugs without exposing real patients to risk. Advances in stem cell biology and tissue engineering are inching us closer to bioengineered organs for transplantation, or at least lab-grown tissues that can help repair damage after heart attacks or spinal cord injuries. On the genetic front, more precise editing tools and safer delivery methods are being developed to correct inherited disorders at their source rather than just managing symptoms.
Immunologists are refining next-generation immunotherapies that could make certain cancers manageable chronic conditions or even prevent them in high-risk groups. At the same time, wearable sensors and noninvasive imaging are expanding the ways we can monitor physiology in real time, turning everyday life into a continuous health study if people choose to opt in. Yet these possibilities carry heavy ethical and social questions: who gets access, how data are protected, and whether interventions might be used to enhance as well as heal. The future landscape of human-body science is not just a technical puzzle but a moral one, and decisions made over the next decade will shape whether these astonishing tools narrow or widen existing gaps in health and opportunity.
10. How You Can Engage With the New Science of Your Own Body
It’s easy to feel like these discoveries belong only in high-tech labs or specialist clinics, but there are simple ways to engage with this new understanding of the body in everyday life. Paying attention to habits that support your microbiome – such as eating a variety of fiber-rich foods, being cautious with unnecessary antibiotics, and spending time outdoors – aligns with what many studies suggest about microbial diversity and resilience. Respecting your internal clocks by prioritizing regular sleep, dimming bright screens at night, and keeping meal times relatively consistent can support the metabolic and hormonal rhythms that these discoveries have brought into focus. Small changes in stress management, from brief daily walks to mindfulness exercises, can help calm the brain–body feedback loops that otherwise amplify inflammation and strain.
If you’re curious or motivated to go further, you can follow reputable science reporting, support organizations that fund medical and physiological research, or even participate in citizen science and clinical research studies when appropriate. Asking questions at medical appointments about personalized approaches – such as whether a treatment is informed by genetic, microbial, or lifestyle factors – helps push the healthcare system to fully absorb these breakthroughs. Above all, staying curious about your own body, rather than viewing it as a black box, is a powerful form of engagement. The story of human physiology is still being written, and each of us is living evidence of how surprising that story can be.

Suhail Ahmed is a passionate digital professional and nature enthusiast with over 8 years of experience in content strategy, SEO, web development, and digital operations. Alongside his freelance journey, Suhail actively contributes to nature and wildlife platforms like Discover Wildlife, where he channels his curiosity for the planet into engaging, educational storytelling.
With a strong background in managing digital ecosystems — from ecommerce stores and WordPress websites to social media and automation — Suhail merges technical precision with creative insight. His content reflects a rare balance: SEO-friendly yet deeply human, data-informed yet emotionally resonant.
Driven by a love for discovery and storytelling, Suhail believes in using digital platforms to amplify causes that matter — especially those protecting Earth’s biodiversity and inspiring sustainable living. Whether he’s managing online projects or crafting wildlife content, his goal remains the same: to inform, inspire, and leave a positive digital footprint.