Have you ever wondered why some people seem to age gracefully while others show the wear of years much earlier? Your body carries invisible timekeepers that count down the moments of your existence at the cellular level. These aren’t the clocks on your wall or the birthdays on your calendar. They’re far more intricate and, honestly, a bit unsettling when you really think about what they mean for your future.
The biological mechanisms governing aging have puzzled scientists for decades. While you celebrate each passing year, your cells are undergoing profound changes that gradually erode your vitality. Let’s dive in to explore the remarkable systems operating inside you right now, quietly determining how fast or slow you’ll age.
Telomeres: Your Cellular Countdown Timer
Think of telomeres as the plastic tips on your shoelaces. These repeated DNA sequences appear at the end of your chromosomes and act as a biological clock for each cell, counting the number of times a cell has divided. Every time your cells replicate to replace old or damaged tissue, these protective caps get slightly shorter.
Each time a cell undergoes division to create a new cell, its telomere shortens, and eventually the chromosome’s telomere becomes too short to divide further, triggering the cell into a state of senescence. This isn’t just theoretical science happening in a laboratory dish. Telomere shortening and damage are recognized causes of cellular senescence and aging, affecting every tissue in your body from your skin to your heart.
What’s particularly fascinating is that telomere length isn’t entirely predetermined. People with stressful lives have been found to have telomeres that are shorter than average, while a study showed that people who adopted healthier lifestyles experienced on average a 10 percent increase in telomeres. This means the choices you make today might influence the speed of your biological clock tomorrow.
DNA Damage: The Accumulating Burden

Your DNA takes a beating every single day. It is estimated that our DNA is damaged up to a million times a day. Let that sink in for a moment. While you’re reading this sentence, thousands of microscopic assaults are occurring within your cells.
Most of this damage is repaired immediately because cells have efficient detection and repair mechanisms, however these repair processes are not perfect and a small percentage of damage remains unrepaired, which is why DNA damage accumulates as we age. Inherited defects in these maintenance systems not only predispose to cancer but also underlie numerous forms of premature aging in humans, indicating a tight link between genome integrity, cancer and aging.
The real kicker here is that you can’t entirely avoid DNA damage. Everyday functions like breathing and moving create highly unstable molecules called free radicals, though external factors such as UV radiation and pollution can create an excess of these, causing damage to molecules in cells and DNA. So even living your normal life contributes to your aging process, though exposure to environmental toxins can certainly accelerate it.
Mitochondrial Decline: Your Energy Crisis
Mitochondrial dysfunction significantly impacts aging because mitochondria regulate cellular energy, oxidative balance, and calcium levels. These tiny powerhouses inside your cells don’t just produce energy – they’re central players in determining how well you age.
As you grow older, these cellular batteries start to fail. With advanced age, mitochondrial DNA volume and functionality decrease due to accumulation of mutations and oxidative damage, and mitochondria are characterized by impaired function such as lowered oxidative capacity, reduced oxidative phosphorylation, decreased ATP production, and significant increase in ROS generation.
Here’s where things get interesting. Current evidence shows that mitochondrial DNA mutations and oxidative damage do not causally limit physiologic aging, instead an evolving literature links age-related loss of mitochondrial signaling and function to important physiologic changes of aging. What matters isn’t just the damage itself, but how your mitochondria communicate distress signals throughout your body.
The Epigenetic Clock: Reading Your Biological Age

Your biological age might be quite different from the number on your driver’s license. An epigenetic clock is an analytical method used as a biomarker of aging to estimate biological age, relying on age-related modifications to DNA that occur over time, with many clocks based on the analysis of DNA methylation.
Scientists can now examine specific chemical modifications to your DNA and calculate how old your body really is at the molecular level. Surprisingly, the DNA methylation pattern of only 350 methylation sites is sufficient to predict a person’s biological age, and this epigenetic clock has become an important tool as a biomarker.
What’s truly remarkable is the predictive power of these clocks. A higher biological age – regardless of chronological age – consistently predicted an earlier death. Research discovered that roughly five percent of the population ages at a faster biological rate, resulting in a shorter life expectancy, with accelerated aging increasing risk of death by fifty percent at any age. This might explain why some seemingly healthy individuals die unexpectedly young despite doing everything right.
Cellular Senescence: When Cells Refuse to Die
Some of your cells eventually reach a point where they can no longer divide, yet they refuse to die. Senescence refers to the aging process on a cellular level, and as we age, more cells lose the ability to divide, with the accumulation of senescent cells leaving lasting impacts from the appearance of wrinkles to the emergence of age-related health conditions.
These zombie cells don’t just sit there quietly. Physiologically, senescence acts as a safeguard against tumorigenesis by preventing the proliferation of damaged cells, however persistent activation of senescence can contribute to various pathological conditions, particularly those associated with aging, cancer, and chronic diseases. It’s a double-edged sword – your body’s attempt to prevent cancer may be accelerating other forms of aging.
The inflammatory substances these senescent cells release can damage surrounding healthy tissues. Senescence is a process leading cells to the SASP phenotype, and permanent senescence of many SASP cells contributes to faster aging of the body and development of age-related diseases due to release of pro-inflammatory factors. Essentially, these cells are poisoning your tissues from within.
Oxidative Stress: The Free Radical Theory
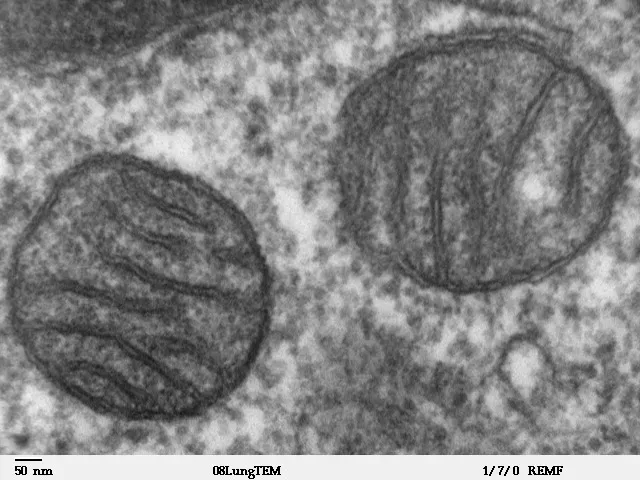
For decades, scientists believed free radicals were the primary villains in the aging story. Mitochondria are the main source of reactive oxygen species produced as a by-product of mitochondrial respiration, and these free radicals can damage macromolecules such as DNA, lipids and proteins, and for a long time ROS were thought to be the main culprits in the ageing process.
The picture is now more nuanced. Recent studies challenge this view and suggest that ROS may instead act as signalling molecules within the cell, and in some ways increased levels of ROS may even be beneficial, activating cellular defence and repair mechanisms. This explains why taking massive doses of antioxidants doesn’t necessarily slow aging – the story is far more complicated than simply neutralizing free radicals.
The Hallmarks of Aging: A Complex Web
Aging doesn’t result from a single cause. Primary hallmarks include genomic instability, telomere attrition, epigenetic alterations, and loss of proteostasis, while antagonistic hallmarks emerge as compensatory mechanisms including deregulated nutrient sensing, mitochondrial dysfunction and cellular senescence, which when they fail lead to integrative hallmarks such as stem cell exhaustion and chronic inflammation.
These mechanisms don’t operate independently – they interact in ways scientists are only beginning to understand. Research suggests that human aging accelerates midlife rather than progressing steadily throughout adulthood, and understanding this shift could help guide earlier and more targeted approaches to support healthier aging.
The mechanisms of aging we currently understand may not be the same ones that set the upper boundary for lifespan. There may be fundamental limits we haven’t yet discovered, biological barriers that determine the maximum human lifespan regardless of how well we understand or manipulate the known aging processes.
Where Science Meets Your Future
The knowledge emerging from aging research carries profound implications for how you might approach your own health. Many longevity pathways identified in worms, flies and mice are relevant to humans, though we are at the beginning of true translation. Human clinical trials are just beginning, enabled by the invention of multiomics biomarkers of aging, and more basic research is needed to develop mechanism-based interventions.
The reality is that roughly two-thirds of all human deaths are now caused by aging-related processes. Aging is among the greatest known risk factors for most human diseases, and of the roughly 150,000 people who die each day across the globe, about 100,000 die from age-related causes, with the proportion reaching 90 percent in industrialized nations. This makes understanding and potentially intervening in the aging process not just a scientific curiosity but a pressing medical necessity.
The biological clocks ticking inside you aren’t entirely fixed. Your lifestyle choices, environmental exposures, and perhaps soon, targeted medical interventions, all influence how fast these timekeepers run. The science of aging has moved from asking whether we can slow the process to determining how we might do so safely and effectively. What remains uncertain is how quickly these discoveries will translate into treatments that extend not just lifespan, but the years of healthy, vibrant living. What choices will you make knowing these clocks are counting down every moment?

Hi, I’m Andrew, and I come from India. Experienced content specialist with a passion for writing. My forte includes health and wellness, Travel, Animals, and Nature. A nature nomad, I am obsessed with mountains and love high-altitude trekking. I have been on several Himalayan treks in India including the Everest Base Camp in Nepal, a profound experience.