Every person who has ever looked in a mirror and noticed the slow creep of fine lines or felt the morning stiffness that wasn’t there a decade ago has wondered, at least briefly, what is actually happening inside. Aging feels deeply personal, yet it unfolds according to mechanisms that are almost universal across all living things. It’s not simply the passage of time that wears the body down – it’s a cascade of biological processes happening quietly at the cellular level, often for decades before their effects become visible.
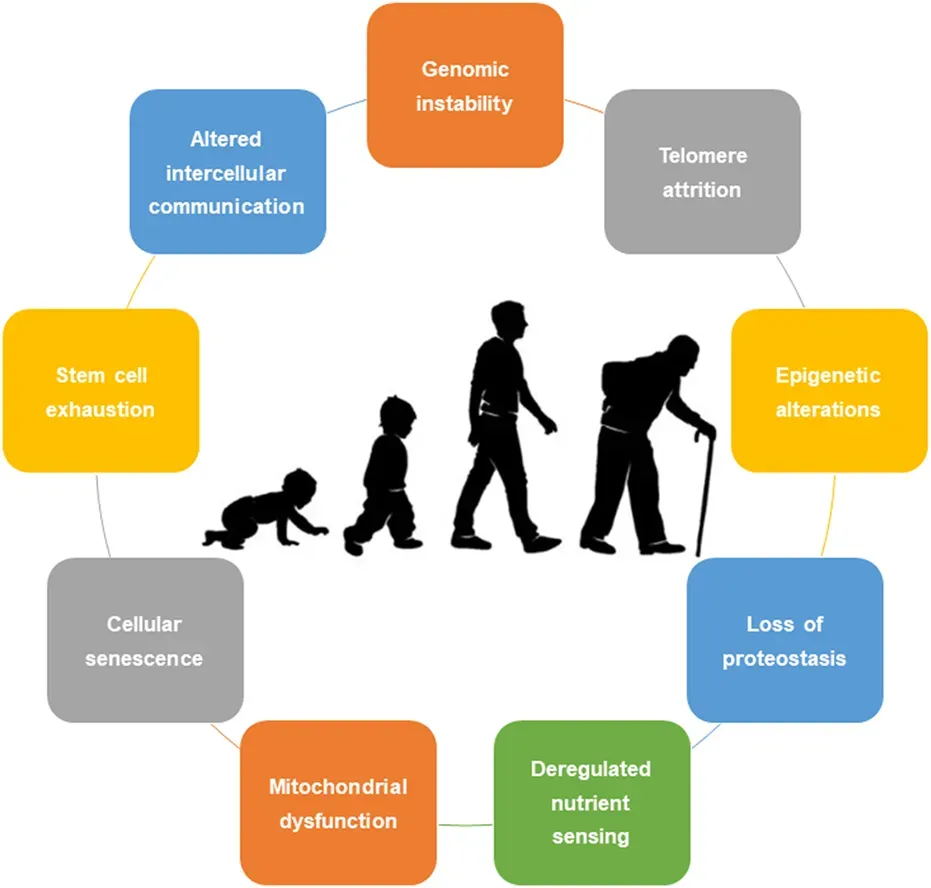
Aging is a complex biological process characterized by a gradual decline in cellular and physiological function, involving a set of interconnected mechanisms that scientists now call the hallmarks of aging, including genomic instability, telomere attrition, epigenetic alterations, mitochondrial dysfunction, and cellular senescence. Understanding these mechanisms doesn’t just satisfy scientific curiosity – it opens real possibilities for slowing the process, managing its consequences, and living longer in better health.
The Hallmarks of Aging: A Biological Framework

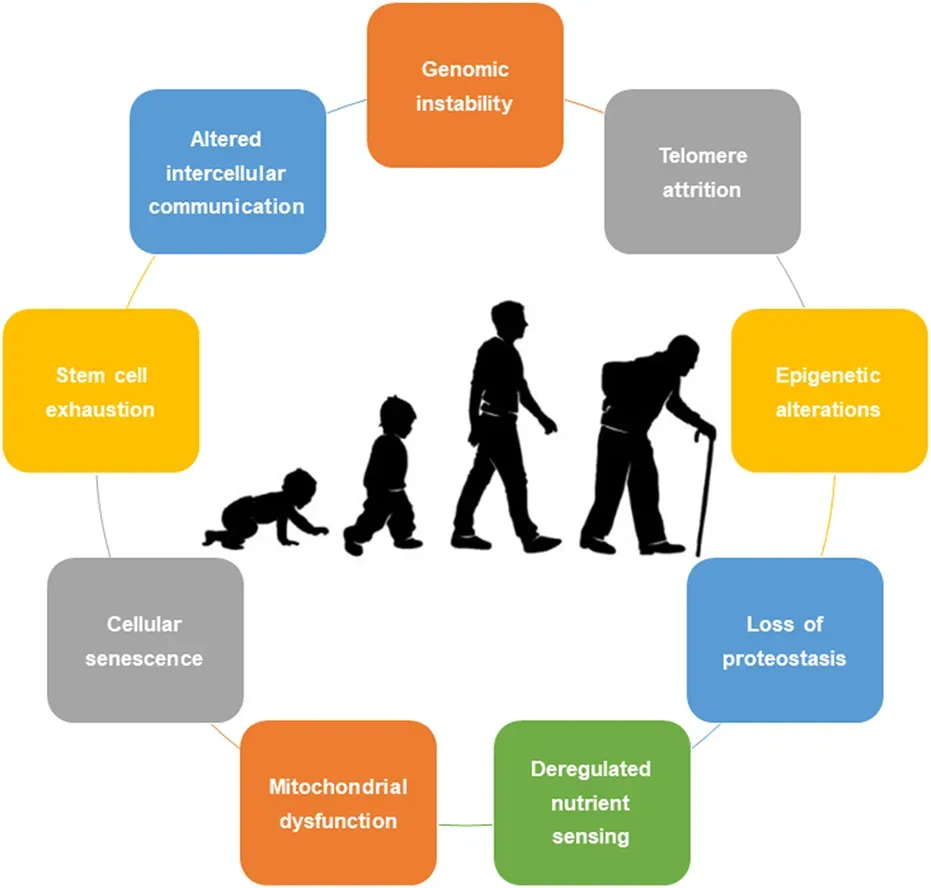
Scientists haven’t always had a shared language for describing how aging works. That changed in 2013 when a landmark framework identified the core biological processes that collectively drive age-related decline. In that framework, aging is organized around twelve biological processes, including genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, disabled macroautophagy, deregulated nutrient-sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, altered intercellular communication, chronic inflammation, and dysbiosis.
The hallmarks can be understood as three interconnected layers. Primary hallmarks, such as genomic instability, telomere attrition, and epigenetic alterations, reflect the accumulation of molecular and cellular damage over time. In response, antagonistic hallmarks emerge as compensatory mechanisms, including deregulated nutrient sensing, mitochondrial dysfunction, and cellular senescence. When these fail or become deleterious, they lead to integrative hallmarks, such as stem cell exhaustion, chronic inflammation, and altered intercellular communication, which drive systemic aging and functional decline.
Telomeres: Your Chromosomes’ Shrinking Timekeepers
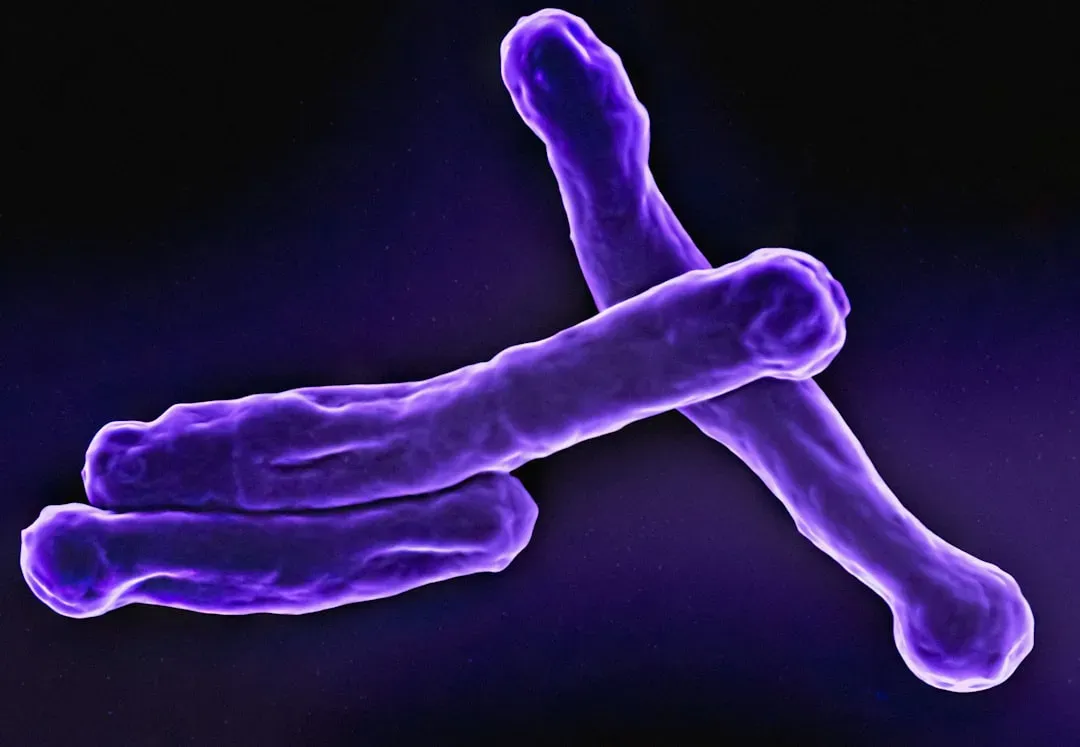
Telomeres, the protective caps at the ends of chromosomes, play a crucial role in maintaining genomic stability and cellular lifespan. As cells divide, telomeres progressively shorten, eventually leading to cellular senescence or apoptosis. Think of them as the plastic tips at the end of a shoelace – without them, the underlying genetic material starts to fray and unravel.
Each time a cell divides, a piece of telomere is lost, so the more cells divide and the older we get, the shorter the chromosome ends become. When a certain length is reached, cells enter a resting phase and stop dividing. These cells can then die, or they can develop into a senescent cell. Beyond simple wear, while repeated rounds of cell division cause telomere attrition, other factors influence the rate at which telomeres shorten – multiple studies have shown that mild oxidative stress can accelerate the rate of telomere shortening, reducing the cells’ ability to proliferate and inducing premature cellular senescence.
Cellular Senescence: When Cells Refuse to Die
Cellular senescence is an evolutionarily conserved process that leads to an irreversible arrest of the cell cycle. It is triggered by various forms of cellular stressors, including DNA damage, telomere shortening, oxidative stress, and oncogene activation, in an effort to halt the proliferation of defective cells – serving to prevent malignant transformation or a switch to apoptosis. The body essentially puts these damaged cells in a kind of permanent pause, which is useful in the short term for preventing cancer.
Cellular senescence is a major factor in the biology of aging, where damaged cells stop dividing but do not die. These “zombie cells” remain active in the body and begin to release harmful signals that affect nearby healthy cells, and their buildup contributes to inflammation and tissue decline over time. As you age, more cells lose the ability to divide, and the number of senescent cells in your body increases – and their accumulation leaves lasting impacts on the overall aging process, from the appearance of wrinkles to the emergence of age-related health conditions.
The Epigenetic Clock: Reading Your Biological Age
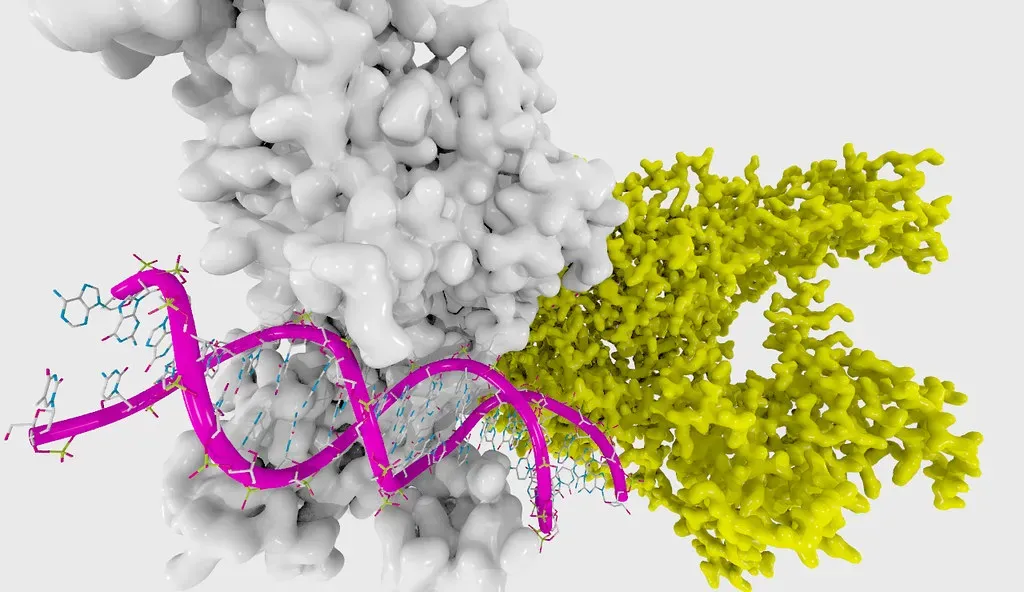
An epigenetic clock is an analytical method used as a biomarker of aging to estimate biological age. The method relies on age-related modifications to DNA that occur over time and regulate how genes are expressed, with many epigenetic clocks based on the analysis of DNA methylation, measuring the accumulation of methyl groups to specific regions of DNA molecules. In other words, your DNA carries a running record of how much biological wear and tear your cells have accumulated, independent of your actual birthday.
Scientists can now measure biological age using patterns of chemical tags on DNA called DNA methylation – these “epigenetic clocks” estimate how old your body actually is, independent of your birth date. Two people born in the same year can have biological ages that differ by a decade or more. When your predicted biological age exceeds your chronological age, you’re experiencing epigenetic acceleration, which correlates with higher risks of disease and earlier death. This gap is shaped by genetics, but your lifestyle choices influence it considerably as well.
Mitochondrial Dysfunction: When the Cell’s Power Plants Fail
Mitochondria are small organelles in the cell that are not only the cellular power plants but also form a central hub for metabolic processes. They use oxygen to produce energy in a process called mitochondrial respiration, and an important feature of mitochondria is that they contain their own DNA, called mtDNA, which codes for the proteins needed for respiration. This makes them uniquely vulnerable – their own genetic material can accumulate mutations over time, unlike the tightly protected DNA in the cell nucleus.
Mitochondria are also the main source of reactive oxygen species (ROS), which are produced as a by-product of mitochondrial respiration. These free radicals can damage other macromolecules such as DNA, lipids, and proteins, and are therefore potentially harmful to the cell. Mitochondrial dysfunction is a key aspect of the biology of aging – mitochondria are responsible for generating energy in cells, but their performance decreases over time, leading to reduced energy availability, which can affect everything from physical strength to cognitive performance.
Inflammaging: The Slow Burn of Chronic Inflammation
Aging is associated with chronic, low-grade inflammation, commonly called inflammaging. Aging affects your immune system, leading to a decline in its function and increased inflammation, known as immunosenescence and inflammaging. Unlike the acute inflammation you feel when you cut your finger – which resolves quickly and serves a clear purpose – this background inflammation smolders for years without an obvious trigger, quietly damaging tissues and organs.
Factors secreted by senescent cells, known as the senescence-associated secretory phenotype (SASP), promote chronic inflammation and can induce senescence in normal cells. At the same time, chronic inflammation accelerates the senescence of immune cells, resulting in weakened immune function and an inability to clear senescent cells and inflammatory factors, which creates a vicious cycle of inflammation and senescence. Immunosenescence and inflammaging are mutually reinforcing processes: age-associated immune dysfunction fuels chronic inflammation, which in turn accelerates immune aging, establishing a self-perpetuating cycle.
Genetics, Evolution, and Why Aging Even Exists
It’s worth stepping back and asking a broader question: why does aging exist at all? From a biological standpoint, it seems counterproductive. Theories about why we age can be grouped into two camps: gradual damage over time and genetic programming. The answer from evolutionary biology is that aging evolved not because it is beneficial, but as a side effect of something else.
George Williams’ theory of antagonistic pleiotropy states that natural selection can favor gene variants with beneficial effects early in life, even if the same variants have deleterious effects later. Because the deleterious effects of these genes occur in old age, after the reproductive phase, they have little evolutionary impact. Nature cannot directly select against a gene or its mutation that causes the death of an individual in old age if its deleterious effects do not occur before the end of the reproductive phase. In other words, once your genes have done their reproductive job, evolution has little incentive to maintain your body indefinitely.
Your genes are one of the main components that impact how and why you age, with your unique genetic combinations playing a role in determining your overall lifespan. Some individuals are born with robust cellular repair mechanisms, which allows them to physically age at a slower rate than those that have less efficient DNA repair mechanisms.
Conclusion: The Clock Ticks Differently for Everyone

Aging is not a single event. It’s the compounding result of dozens of biological processes, each nudging the body gradually away from its youthful equilibrium. What makes aging feel fast is that these processes don’t operate in isolation. Mitochondrial damage increases free radicals, which accelerate DNA mutations, which produce more senescent cells, which drive inflammation, which impairs immune function, which allows even more senescent cells to accumulate – each mechanism feeds into others, and the pace of decline compounds over time.
This process is highly context-dependent. Everyone ages differently because each individual is unique in terms of genetics and life history, a concept sometimes called the “exposome,” which is the measure of all the exposures of an individual in a lifetime. Choices around diet, sleep, exercise, and stress don’t just affect how you feel today – they leave measurable biological marks on your cells that influence how quickly those clocks tick.
The science of aging has come a long way from simply accepting the process as inevitable decay. What researchers now understand is that aging is biological, measurable, and at least partially modifiable. The biological clocks inside your body are real – and learning to read them may be one of the most important things you can do for your long-term health.