Somewhere around your late twenties, something quietly changes. You still feel like the same person, but your cells start moving from growth and repair into a slow, steady decline. You do not notice it at first, then one day you catch your reflection, or feel your knees on the stairs, and you suddenly realize: time has been doing its work all along.
For most of history, aging felt like a mysterious curse: inevitable, unstoppable, and poorly understood. Now, for the first time, scientists are not just asking why we age, but how we might slow it down. The surprising part is that aging is turning out to be less of a simple countdown and more of a complex, programmable system – with weaknesses, levers, and maybe even off-switches we’re just beginning to find.
The Surprising Truth: Aging Is Not Just “Wear and Tear”

It used to sound comforting to say our bodies age the way a car does: parts wear out, and eventually they stop working. But that metaphor turns out to be only half true and, honestly, a bit lazy. Biologists now see aging as a set of specific, measurable processes that go wrong over time, not just random damage piling up without any pattern or rules. In other words, it is less a rusty car in the rain and more a complex software system slowly glitching line by line.
This shift matters because what we can understand, we can often change. Studies in worms, flies, mice, and even primates have shown that you can extend lifespan dramatically by tweaking certain genes, altering diet, or adjusting cell signaling pathways. Some animals barely seem to age at all, and that blows up the idea that aging must always look the same. Aging is not a single switch that flips at a certain birthday; it is a collection of dials, and researchers are learning which ones we might realistically turn.
DNA Damage and Epigenetic Clocks: Time Written in Our Cells

Every day, your DNA is attacked by sunlight, chemicals, and normal metabolism, and your cells scramble to repair the damage. Most of the time, they manage, but not perfectly. Over decades, small mistakes accumulate like typos in a long book, and some of those mistakes affect genes that keep cells healthy, controlled, and functional. This gradual buildup of damage makes cells more prone to malfunction, cancer, or death.
On top of that, there is another layer: epigenetics, the chemical tags that tell your genes when to turn on or off. These tags shift predictably as we age, so predictably that scientists built “epigenetic clocks” that can estimate your biological age from a simple blood sample. The wild part is that in some experiments with animals, and early pilot work in humans, certain interventions seem to slightly “turn back” these clocks. It is still early and far from a magic reset button, but it proves something powerful: the aging process is written in code, and parts of that code may be editable.
Cellular Senescence: Zombie Cells That Refuse to Die
As cells divide over and over, some reach a point where they stop dividing but do not die. These are called senescent cells, and they are like unwanted guests who never leave the party and then complain about the music. At first, this process is protective: it prevents damaged cells from becoming cancerous. But with age, senescent cells accumulate in tissues all over the body, leaking inflammatory chemicals that harm nearby healthy cells.
In mouse experiments, removing these “zombie” cells using special drugs called senolytics led to healthier, longer-lived animals with better physical function. This finding ignited massive interest because it suggests that just clearing out a specific type of malfunctioning cell could slow some aspects of aging. Early human trials are cautious and tightly controlled, but they are happening now. Imagine a future where part of your midlife health checkup involves flushing out senescent cells the way we treat clogged arteries today.
Mitochondria and Metabolism: The Energy Crisis Inside Your Cells
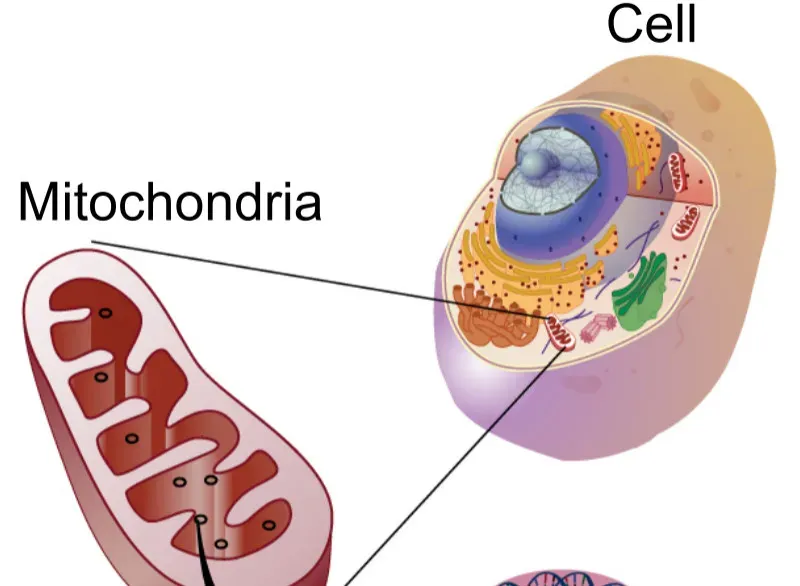
Mitochondria are the tiny power plants inside your cells, turning food into usable energy. When they are young and efficient, you feel it: stable energy, good recovery, mental sharpness. With age, mitochondria tend to become less efficient and more prone to producing harmful byproducts called reactive oxygen species. Over time, this can damage cell components and worsen the very energy systems that keep you alive.
Researchers have linked healthier mitochondrial function with longer healthspan in many organisms, from worms to rodents. Interventions like exercise, intermittent fasting, and certain nutrients appear to nudge mitochondria to recycle and renew themselves, a process called mitophagy. Some experimental drugs and supplements aim to boost mitochondrial performance or repair their DNA directly. If aging is partly an energy management problem, then keeping your cellular power grid resilient could be one of the most realistic levers we have to stay functional for longer.
Inflammaging: When Your Immune System Turns Against You

Inflammation is supposed to be your body’s emergency response team: rush in, fix the problem, get out. With aging, that system misbehaves. The immune system becomes less sharp at fighting infections and cancer, but at the same time, low-level chronic inflammation spreads quietly through the body. Scientists even have a word for it now: inflammaging. It is like having a tiny, invisible fire smoldering in your tissues year after year.
This chronic inflammation is strongly linked to heart disease, diabetes, dementia, frailty, and many other age-related conditions. The sources are varied – senescent cells, excess visceral fat, poor sleep, gut microbiome changes, and more. New treatments are being tested that target inflammatory pathways specifically involved in aging rather than short-term infections. While those drugs are still being studied, habits like regular physical activity, better sleep, and nutrient-dense diets are already proven to dial down chronic inflammation, buying you extra quality years even before high-tech therapies arrive.
Longevity Pathways: mTOR, AMPK, and the Biology of “Eat Less, Live Longer”

One of the most consistent findings in aging research is almost annoyingly simple: in many species, eating fewer calories without malnutrition extends lifespan and healthspan. Under the hood, this is not magic; it is biochemistry. Nutrient-sensing pathways like mTOR, AMPK, and sirtuins act like cellular thermostats, telling your body whether to focus on growth and reproduction or on repair and maintenance. When food is plentiful, the body leans into growth. When calories are lower, the body flips into a repair mode that seems to slow aging.
Drugs like rapamycin and metformin, and molecules that activate sirtuins, are being studied because they tap directly into these pathways. In animals, they often mimic some benefits of calorie restriction, improving health markers and sometimes extending lifespan. No one has a universally accepted “longevity pill” yet, and there is real debate and caution around risks, dosing, and long-term effects in humans. But the core message is clear: your body has built-in programs for survival and conservation, and modern lifestyles constantly push the dial toward growth and overconsumption instead of repair.
Can We Really Slow Aging? What Actually Works Right Now

With all the talk about gene editing, senolytics, and longevity drugs, it is easy to forget an uncomfortable truth: the most reliable, evidence-backed tools for healthy aging in humans are still frustratingly basic. Regular physical activity, not smoking, maintaining a healthy body weight, managing blood pressure and blood sugar, eating mostly whole, minimally processed foods, and sleeping enough hours are repeatedly linked to longer life and fewer years of disability. Large population studies show that people who do several of these at once can add many extra years of relatively good health, not just extra time in a hospital bed.
That might sound boring compared to futuristic lab work, but it is also empowering. You can do something meaningful this week, this month, this year, without waiting for a clinical trial to finish. At the same time, serious work on advanced therapies is moving fast, from partial cellular reprogramming that may reset aging markers, to personalized medicine guided by biological age tests instead of just your birthday. It is becoming more realistic to imagine a future where turning seventy feels more like today’s fifty, and where dying young of an old person’s disease is seen as a preventable failure rather than bad luck.
The Ethics and Future of Longevity: How Long Should We Live?

The question is no longer only “Can we extend life?” but “What kind of life are we extending, and for whom?” If powerful longevity therapies become real, they will raise messy questions about inequality, access, and what it means to age together as a society. A world where only the wealthy can afford extra decades of robust health would deepen divides that already feel unbearable. On the other hand, if these tools are made widely available and affordable, they could shift entire economies, retirement systems, and how we think about work, education, and family.
There is also a quieter, more personal question: how much time is enough if that time is good? Some people dread the idea of living significantly longer, imagining only extended frailty and dependence. But much of modern longevity science is focused less on extreme lifespan and more on healthspan – stretching the healthy, active years and compressing the period of decline at the end. If that vision becomes reality, the biggest change might not be in the number of candles on the cake, but in how many of those years you spend actually feeling like yourself.




