In quiet labs filled with humming incubators and robot arms, researchers are attempting something that once lived only in science fiction: crafting living systems from the ground up. The goal isn’t to play creator; it’s to understand life so well that we can rebuild it, redesign it, and, when needed, repair it. This push has gathered momentum as DNA writing becomes cheaper and the old trial-and-error playbook of bioengineering hits its limits. The mystery is simple to ask and hard to answer: how few parts does life need, and what could we do if those parts were fully programmable? The stakes span medicine, climate, and security, which is why this story now reads less like a distant dream and more like a developing beat in modern science.
The Hidden Clues
What if life can be stripped to a handful of instructions and still run? That question has driven teams to assemble “minimal cells,” organisms pared down to the smallest working genome so we can see which genes truly matter. The surprising twist is that even after extreme streamlining, roughly about one third of the genes that remain essential still have functions we don’t completely understand.
I remember standing in a microscopy room during a lab tour, watching a minimal bacterium wobble, divide, and steady itself like a newborn fawn. It looked so simple and yet carried secrets that textbooks still gloss over. Those hidden functions are the breadcrumbs, and following them is helping scientists map the non-obvious rules that make biology tick.
From Ancient Tools to Modern Science

Humanity has been editing life since we fermented dough and domesticated crops, but those were blunt tools compared with the precision now on tap. Today’s labs blend genome editing, rapid DNA synthesis, and automated testing to cycle through designs far faster than traditional biology allowed. Instead of nudging nature and waiting, researchers sketch a blueprint, write the DNA, and test the result in days.
This shift mirrors the leap from handwritten ledgers to spreadsheets: the job is the same, the scale is not. With robotic liquid-handlers and cloud-linked data pipelines, hypotheses aren’t just tried – they’re stress-tested by the hundreds. The pace doesn’t just accelerate discovery; it changes the kinds of questions we can afford to ask.
Stitching Genomes Like Code
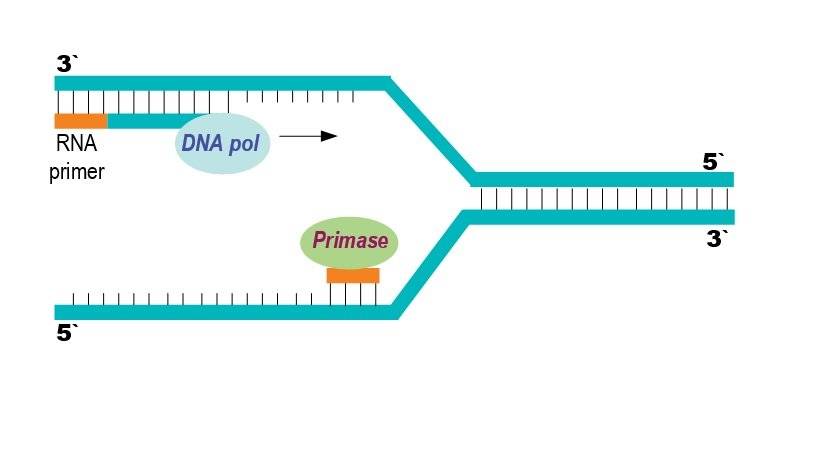
Building synthetic life starts with writing long stretches of DNA, then stitching them together into whole chromosomes that cells can read. The cost of DNA synthesis has fallen dramatically over the past two decades, turning projects that once seemed extravagant into routine lab work. That means scientists can redesign genomes – recoding the genetic alphabet, removing redundancies, or inserting safety features – much like refactoring software.
Some teams go further, exploring “xenobiology,” where organisms use expanded genetic alphabets or nonstandard biochemical parts. The idea is to create life that’s both useful and less likely to mingle with natural ecosystems, like a power tool that only works in a dedicated outlet. In practice, that can mean resistance to viruses, reduced gene-sharing with wild microbes, and cleaner control over what a cell can or cannot do.
The Minimal Cell, Revisited

Milestones in the field include the first bacterial cell controlled by a synthetic genome and later a radical simplification that removed hundreds of genes while keeping the organism viable. Those efforts weren’t just stunts; they were stress tests that revealed how many backup systems and hidden dependencies lurk inside even the smallest life forms. Researchers then tweaked these cells to fix awkward growth and division, showing that a stripped-down organism can be tuned like a lean engine.
What’s fascinating is how these minimal cells become blank canvases for learning and invention. Add a module for producing a medical enzyme, and you’ve got a hyper-focused microfactory; add a circuit for sensing toxins, and you have a biological alarm. Each tweak is an experiment in cause and effect, a way to turn life into an explainable machine rather than a mystical black box.
Why It Matters
Traditional genetic engineering modifies what nature gives us, which often behaves like a used car with unknown quirks; synthetic life aims to build the chassis ourselves and document every bolt. That switch matters when reliability is nonnegotiable, whether you’re making vaccines, producing rare drugs, or growing sustainable materials. Fewer unknowns in the genome mean fewer surprises in the factory and clinic.
There’s also a security angle that rarely makes headlines but quietly shapes the field. By recoding genomes and adding containment features, designers can reduce the risk of cross-talk with wild organisms or viral hijacking. If biology is going to be the manufacturing platform of the century, predictability and safeguards can’t be afterthoughts – they’re the operating system.
The Hidden Clues in Health and Environment
Think of synthetic cells as customizable apprentices: they learn one skill and do it well. In hospitals, that might be living therapeutics that patrol the gut, detect inflammation, and release medicine on cue. In industry, it’s cleaner production of essential chemicals that now rely on fossil fuels, swapping smokestacks for bioreactors.
Environmental labs are also exploring organisms that digest waste streams or capture carbon with fewer side effects. Because their genomes are built, not borrowed, these cells can be designed to depend on lab-made nutrients or to self-destruct outside controlled settings. It’s the biological equivalent of a tether, keeping power and responsibility in the same place.
Global Perspectives

This is a genuinely international story, with research hubs in North America, Europe, and Asia pushing different pieces of the puzzle. Some groups prioritize foundational science – mapping essential genes and cellular logistics – while others drive applications in medicine, agriculture, or materials. Public agencies and private funders both see the momentum, and investment follows the promise of faster discovery and domestic manufacturing.
But speed invites scrutiny, as it should. Ethical review, transparent risk assessment, and community engagement are moving from side notes to center stage in project planning. The balance to strike is open progress with responsible guardrails, so benefits aren’t reserved for a select few while risks are shared by everyone.
The Future Landscape

Over the next decade, expect genome foundries to look more like high-throughput chip fabs, turning digital designs into living prototypes overnight. As DNA writing costs continue to fall, scientists will iterate on whole genomes the way engineers version-control code, rolling updates into safer, smarter cells. Built-in safety switches, metabolic “circuit breakers,” and genetic firewalls will become standard rather than exotic.
We’ll also see hybrids of cell-free systems and minimal cells – biological workhorses that do specific tasks without the baggage of full life. That could shrink supply chains for critical medicines and enable on-demand manufacturing closer to patients and farms. The headline risk is hype; the headline reward is a more robust, resilient bioeconomy that’s harder to break when the world jolts.
How You Can Engage

Start locally: attend public science forums at universities or community labs, ask questions, and learn how researchers weigh risks and benefits. Support hands-on biology education in schools, because tomorrow’s safeguards – and breakthroughs – will be built by students who understand both the code and the consequences. When you see a biotech product on a shelf or in the news, look for clear safety claims and transparent testing, and reward companies that publish methods plainly.
If your city debates bioscience initiatives, show up and ask for practical guardrails like containment practices, third-party audits, and worker training. Consider donating to programs that broaden access to biotech education so the future isn’t designed by and for a narrow slice of society. Wouldn’t you rather help write the instructions for the next era of biology than read them after the fact?

Suhail Ahmed is a passionate digital professional and nature enthusiast with over 8 years of experience in content strategy, SEO, web development, and digital operations. Alongside his freelance journey, Suhail actively contributes to nature and wildlife platforms like Discover Wildlife, where he channels his curiosity for the planet into engaging, educational storytelling.
With a strong background in managing digital ecosystems — from ecommerce stores and WordPress websites to social media and automation — Suhail merges technical precision with creative insight. His content reflects a rare balance: SEO-friendly yet deeply human, data-informed yet emotionally resonant.
Driven by a love for discovery and storytelling, Suhail believes in using digital platforms to amplify causes that matter — especially those protecting Earth’s biodiversity and inspiring sustainable living. Whether he’s managing online projects or crafting wildlife content, his goal remains the same: to inform, inspire, and leave a positive digital footprint.