There is something quietly unsettling about a glass of water. It looks simple enough. Transparent, tasteless, utterly ordinary. Yet scientists have been arguing fiercely for decades about whether this humble molecule might be doing something that defies everything we thought we knew about chemistry. Some have staked careers on it. Others have lost theirs. The debate is still raw in 2026, and honestly, it shows no signs of cooling down.
What if water could hold onto the memory of a substance long after that substance has vanished from it completely? It sounds like science fiction. It sounds like something from a late-night conspiracy forum. Yet Nobel Prize winners have taken the idea seriously enough to publish papers about it, and respected journals have been forced to reckon with the question. So before you dismiss it, let’s dive in.
The Strange and Wonderful Physics of an “Ordinary” Molecule
Let’s be real about something: water is far stranger than most people give it credit for. Water has long been known to exhibit many physical properties that distinguish it from other small molecules of comparable mass, though chemists note that these so-called anomalous properties are entirely predictable consequences of how oxygen’s nuclear charge distorts the electronic clouds of surrounding atoms. That’s the official line, anyway. The reality is that water continues to surprise researchers.
Water is one of the few substances that expands upon freezing, which is why icebergs float on the ocean. Think about that for a moment. Nearly every other liquid on Earth contracts when it solidifies. Water does the opposite. This hydrogen bond behavior is responsible for the anomalously high boiling point of water, the stabilization of protein and nucleic acid structures, and key properties of materials like paper, wool, and hydrogels. For such a tiny molecule, it punches ridiculously far above its weight.
The Hidden Architecture: Hydrogen Bonds and Water’s Inner World
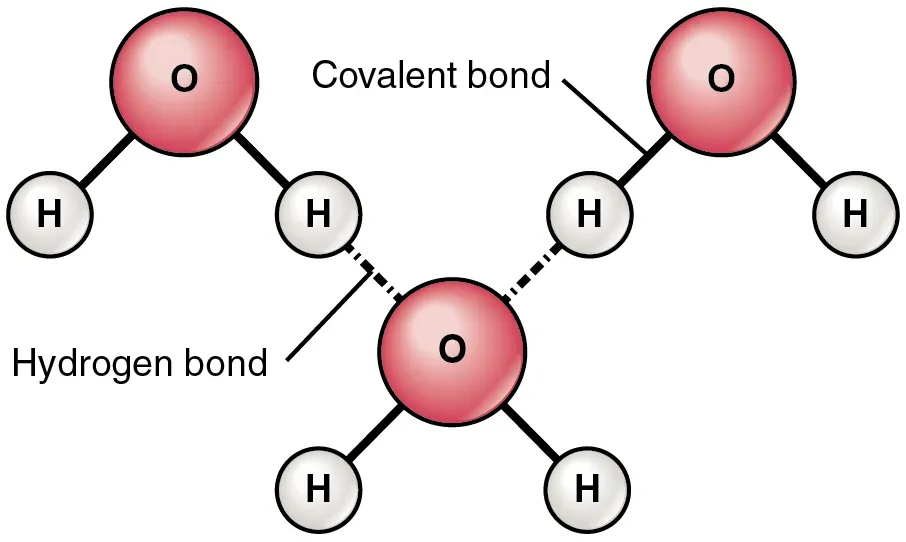
The peculiar properties of water have been ascribed to its intermolecular interactions, specifically strong and directional hydrogen bonds that determine its three-dimensional structure. These bonds are what make water so biologically vital. They are also, as it turns out, at the very heart of the water memory debate.
One factor that makes water unique even among other hydrogen-bonded liquids is its very small mass in relation to the large number of hydrogen bonds it can form, though the lifetime of any single hydrogen bond is very short, on the order of a picosecond. Here’s the thing: a picosecond is one trillionth of a second. The idea that water molecules, connected by hydrogen bonds that last for only about a picosecond before breaking and reforming, could somehow cluster into long-lived mimics of an antibody seemed absurd to many researchers. That tension between fleeting structure and possible lasting effect is exactly where the controversy ignites.
The 1988 Bombshell: Jacques Benveniste and the Paper That Changed Everything

Water memory is a hypothesis that suggests water has the ability to remember substances once dissolved in it, even after those substances have been removed or diluted beyond the point of detection, and it was first proposed by French immunologist Jacques Benveniste in a 1988 paper in the journal Nature, which reported water could retain properties of dissolved biologically active molecules despite extreme dilution. The scientific world did not take this quietly.
Benveniste’s experiments involved diluting an antibody repeatedly until no molecules of the antibody should have been present in the solution, yet the solution allegedly retained the biologic effects on immune cells. Benveniste concluded that the configuration of molecules in water was biologically active, and a journalist coined the term “water memory” for this hypothesis. It was a name that would follow him, and haunt him, for the rest of his life. The publication of their findings in Nature triggered a furore.
Ghostbusters in the Lab: The Nature Investigation

The paper was published in Nature on 30 June 1988 accompanied by an editorial noting that “prudent people should, for the time being, suspend judgement,” and Maddox demanded that the experiments be re-run under the supervision of a hand-picked group that became known as “ghostbusters,” including Maddox himself, famed magician James Randi, and a chemist and freelance debunker at the U.S. National Institutes of Health. It reads like a thriller. A respected scientist, a stage magician, and a fraud investigator all crowded into a laboratory in France.
Further experiments carried out by Benveniste’s team, in double-blind conditions overseen by Maddox, magician James Randi, and fraud investigator Walter Stewart, failed to verify the original results. The next issue of Nature carried a dismissive report headlined “High-dilution experiments a delusion,” the research community reacted with calls for Benveniste’s resignation, and although he did not lose his job, his lab was eventually closed and he was left at loggerheads with the scientific world. It was, by any measure, a spectacular fall from grace.
Undeterred: Digital Biology and the Electromagnetic Turn

At DigiBio, the Paris-based company Benveniste set up in the wake of the controversy, he devised another explanation for his strange results: biomolecules, he said, communicate with their receptor molecules by sending out low-frequency electromagnetic signals, which the receptors pick up like radios tuned to a specific wavelength, and he claimed he was able to record these signals digitally and reproduce their biochemical effect by playing them back to cells. This is the point where many mainstream scientists truly stopped listening.
Around 1991, the transfer of specific molecular signals to sensitive biological systems was achieved using an amplifier and electromagnetic coils, and in 1995, a more sophisticated procedure was established to record, digitize, and replay these signals using a multimedia computer. In the 1990s, Benveniste also asserted that this “memory” could be digitized, transmitted, and reinserted into another sample of water, which would then contain the same active qualities as the first sample. I know it sounds crazy, but the experiments themselves were not entirely without structure, even if the conclusions were deeply disputed.
A Nobel Laureate Enters the Room: Luc Montagnier’s Extraordinary Claims

Montagnier, who shared the Nobel Prize in physiology or medicine in 2008 for the discovery of HIV, stunned many fellow scientists with claims that DNA emits weak electromagnetic waves that cause structural changes in water that persist even in extremely high dilutions. When a Nobel Prize winner starts talking about water memory, you cannot simply look the other way. Benveniste’s findings had been heavily criticized by the scientific community, but were later supported by the research findings of French virologist Luc Montagnier, a respected scientist who shared the Nobel Prize in Physiology or Medicine in 2008 for identifying the AIDS virus.
In 2009, Montagnier and his collaborators published a paper reporting that bacterial DNA can produce an electromagnetic signal transferred through cell culture medium, and his experiment involved diluting a known water sample with HIV DNA down to high dilution levels of ten to the power of negative ten, after which the highly diluted sample was found to emit electromagnetic signals of low frequencies. Using the polymerase chain reaction to amplify DNA, the solution reconstructed in Italy was able to result in DNA being formed, which an independent lab confirmed had roughly ninety-eight percent fidelity to the DNA of the original sequence from France. Those results were intriguing, to say the least.
Why the Scientific Community Remains Deeply Skeptical

Here’s the thing: being intriguing is not the same as being true. In the years after publication, multiple supervised experiments were made by Benveniste’s team, the United States Department of Defense, BBC’s Horizon programme, and other researchers, but no one has ever reproduced Benveniste’s results under controlled conditions. Reproducibility is the bedrock of science. Without it, a result is essentially a rumor.
The credibility of the peer-review system of the journal in which Montagnier’s 2009 papers were published was questioned, as it was a new journal of which Montagnier himself was chairman of the editorial board. Critics described it as “classic pathological science, dredging around in the noise of irreproducible experiments by practitioners whose expertise is not in these fields, in order to support hypotheses that fly in the face of well-established scientific principles.” It’s hard to say for sure where the line falls between genuine frontier science and wishful thinking, but the reproducibility problem is a serious one that cannot be explained away.
The Experimenter Effect and Alternative Explanations

The “memory of water” experiments suggested the existence of molecular-like effects without molecules, and although no convincing evidence of modifications of water specific to biologically-active molecules has been reported, consistent changes in biological systems were nevertheless recorded, leading to a proposed alternate explanation based on classical conditioning of the experimenter. Think of it like this: if you spend years expecting to see a pattern in your data, your brain becomes wired to find it, even when it is not really there.
Researchers concluded that the results of these experiments were related to experimenter-dependent correlations, which did not support the initial “memory of water” hypothesis, though the fact that a signal emerged from background noise at all remained puzzling. Although there is much support for water showing properties that depend on its prior processing, the experimental evidence indicates that such changes are due primarily to solute and surface changes occurring during processing. In other words, something real might be happening, just not what the water memory theorists claim.
What the Future Might Hold: Water Science in 2026 and Beyond

If water memory were proven to exist, it could revolutionize fields ranging from medicine to materials science, and it would challenge our understanding of chemistry and molecular interactions, potentially leading to new treatments and technologies. That potential is impossible to ignore, and it keeps drawing curious minds back to the question. Water is a fascinating substance with properties not encountered in other compounds, and it has been suggested that making use of water’s anomalous properties could lead to exciting applications in engineering, medicine, and physiology, though the origin of these anomalous properties is far from clear, with strong hydrogen bonds being only part of the answer.
From a physical and chemical perspective, these experiments pose a riddle, since it is not clear what mechanism can sustain a water memory of exposure to molecular signals, and from a biological perspective, the puzzle is what nature of imprinted effect in water structure can impact biological function. Recent research into water’s hydrogen bond networks continues to reveal new structural surprises. Water’s surface tension shows a nonlinear temperature dependence including a reentrant increase in the supercooled regime, described as a longstanding puzzle in physical chemistry, which researchers are now unpacking using molecular dynamics simulations. Water keeps finding ways to remind us that we do not fully understand it yet.
Conclusion: The Most Ordinary Mystery in the World

Water memory, as a formally accepted scientific phenomenon, does not currently hold up under rigorous scrutiny. The experiments that started this entire storm remain unreproduced under truly controlled conditions. The mainstream scientific community has not changed its position: water memory is pseudoscientific in nature, it contradicts the scientific understanding of physical chemistry, and is generally not accepted by the scientific community. That is the honest summary.
Yet something nags. The fact that several credentialed scientists, including a Nobel laureate, invested serious effort into this question means it deserves more than a dismissive wave of the hand. The idea of the memory of water arose in the laboratory of Jacques Benveniste in the late 1980s, and decades later the debate was still ongoing, with an increasing number of scientists reporting they had confirmed the basic results. Science, at its best, stays curious. Water, with its bizarre anomalies, its fleeting hydrogen bonds, and its central role in all life on Earth, is not done surprising us. Maybe the most profound scientific discoveries are still hiding in plain sight, right there in your glass. What do you think – is water simply chemistry, or something far stranger waiting to be understood? Share your thoughts in the comments.




