You’ve probably wondered, at some point, why you can’t just stay young forever. Most of us instinctively blame “time” – as if the calendar itself is the culprit, slowly wearing us down like rust on iron. But what if aging isn’t really about time at all? What if everything you thought you knew about why bodies break down is, at best, only half the story?
Science in 2026 is painting a picture that looks radically different from what textbooks described even a decade ago. Researchers from Harvard to Helsinki are overturning old assumptions, revealing a deeply complex web of biological events happening inside your cells right now – some of which might actually be reversible. That’s where things get genuinely exciting. Let’s dive in.
Aging Is Not Inevitable – It Might Be a Disease

Here’s a thought that stops most people cold: what if aging is not an unavoidable fact of life, but a condition with identifiable causes and potential treatments? Leading longevity researcher Professor David Sinclair has stated that aging could soon be treated as a medical condition rather than an inevitable part of life. That’s not wishful thinking from a fringe thinker – it’s a declaration from a professor of genetics at Harvard Medical School, made at the World Governments Summit in Dubai in February 2026.
Aging is a complex biological process characterized by a gradual decline in cellular and physiological function, involving a set of interconnected mechanisms known as the hallmarks of aging, including genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, altered intercellular communication, and dysregulated nutrient sensing. What’s stunning is the sheer number of these mechanisms – and the fact that scientists now believe targeting them directly could change the trajectory of human health entirely.
Your Cells Have a Clock – And It’s Called Epigenetics
Think of your DNA like a massive instruction manual. Your genes are the text on the pages. Epigenetics, though, is the system of sticky notes and highlighted passages that tells the cell which parts of the manual to read and which to ignore. Research shows that compared with genetic information, the loss of epigenetic information plays a core role in the regulation of aging – a conclusion confirmed in various model organisms, including yeast, nematodes, fruit flies, and mice. In other words, it’s not just that your DNA gets damaged. It’s that your cells gradually forget how to read it correctly.
As you age, epigenetic changes accumulate, leading to gradual alterations in gene expression and accelerating the aging process. Over the past decade, a growing number of studies have revealed that progressive changes to epigenetic information accompany aging in both dividing and non-dividing cells, and functional studies in model organisms and humans indicate that these epigenetic changes have a huge influence on the aging process. Honestly, this is one of the most paradigm-shifting findings in modern biology – your body ages partly because it loses access to its own youthful blueprint.
The Telomere Problem: Your Chromosomes Are Running Out of Shoelace Tips
Here’s a great analogy. Imagine the ends of your chromosomes are like the plastic tips on shoelaces – those little caps that stop the lace from fraying. Telomeres are nucleoprotein structures located at the ends of chromosomes, composed of repetitive sequences that serve two essential functions: protecting chromosome termini and preventing the loss of genetic material during DNA replication. Every time your cells divide, those tips get a little shorter. Eventually, when they erode too much, the cell either stops working properly or dies.
DNA damage, oxidative stress, and telomere shortening are the primary triggers of cellular senescence, endowing senescent cells with deregulated metabolism and mitochondrial damage, a senescence-associated secretory phenotype, and arrested cell cycle. Telomere length is found to be associated with the process of aging as well as age-related diseases, and telomere shortening or attrition is an established hallmark of cellular senescence and aging. Shorter telomeres don’t just mean an older cell – they signal a cascade of damaging events that spread throughout your entire body over time.
Senescent Cells: The Zombie Cells Quietly Destroying Your Body
This might be the most unsettling concept in modern aging science. Cellular senescence is a hallmark of aging and a key process influencing healthspan and age-related disease. Once viewed simply as an irreversible cell-cycle arrest, senescence is now recognized as a complex, dynamic state that profoundly shapes tissue function throughout life. While transient senescence supports development, wound healing, and tumor suppression, the chronic accumulation of senescent cells contributes to inflammation, tissue degeneration, and systemic aging. Think of them as zombie cells – they refuse to die but also refuse to do their job, slowly poisoning everything around them.
Senescent cells accumulate in the body with age and secrete harmful substances known as the senescence-associated secretory phenotype, causing chronic inflammation; at the same time, chronic inflammation leads to a decrease in immune system function, known as immunosenescence, which further accelerates the aging process. Cellular senescence and immunosenescence are closely related to a variety of chronic diseases, including cardiovascular diseases, metabolic disorders, autoimmune diseases, and neurodegenerative diseases. The cycle is vicious: more senescent cells create more inflammation, which creates even more senescent cells. It’s like a fire that spreads to its own fuel source.
Mitochondrial Breakdown: When Your Cells’ Power Plants Fail
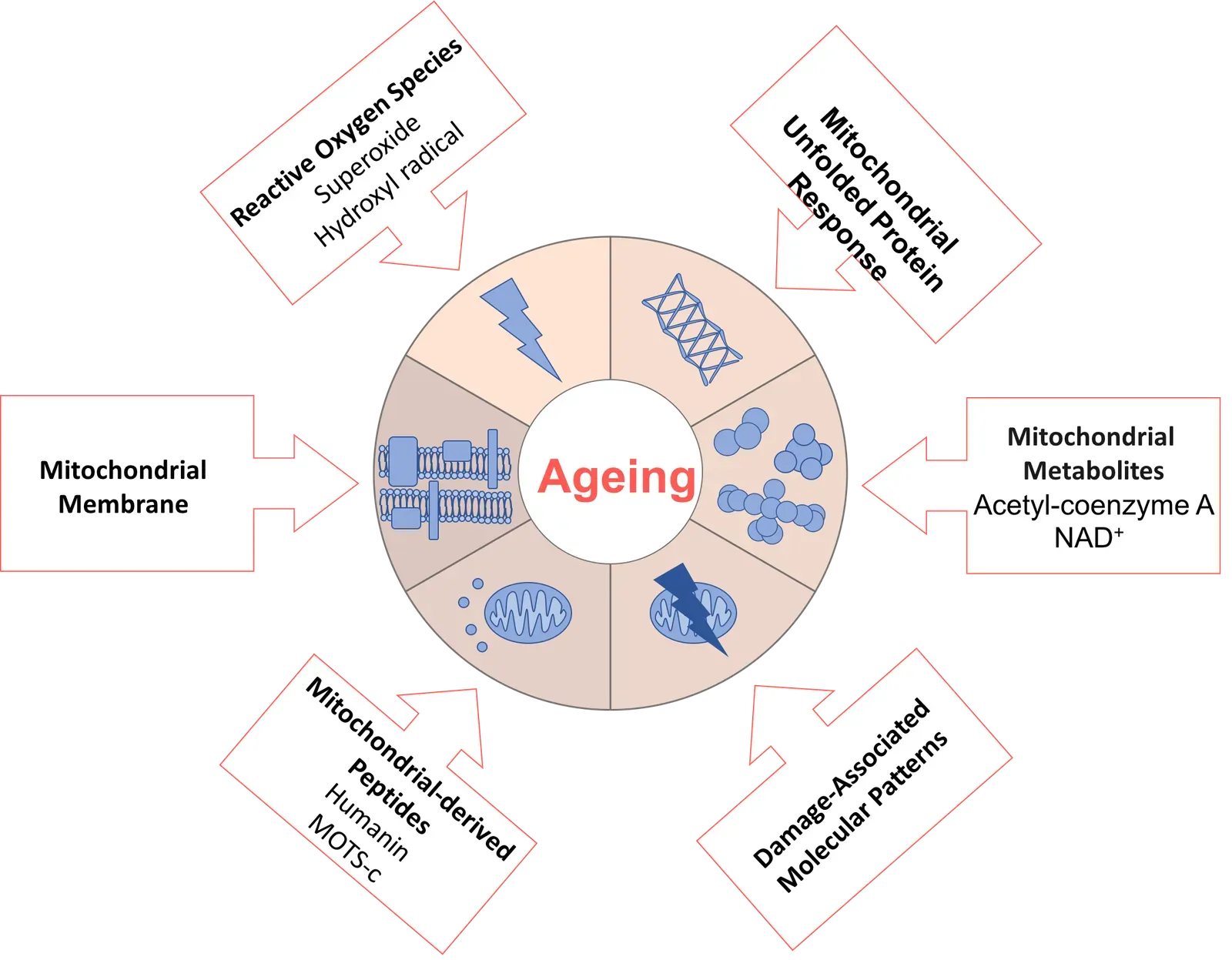
Your mitochondria are the power plants inside every cell, generating the energy that keeps you alive. A series of mitochondrial dysfunctions accompany the aging process, such as reduced oxidative phosphorylation activity, decreased levels of nicotinamide adenine dinucleotide (NAD+) and adenosine triphosphate (ATP), and the accumulation of reactive oxygen species (ROS). When these power plants start malfunctioning, the ripple effects are enormous – and they reach into nearly every tissue in your body.
Mitochondrial dysfunction, characterized by impaired bioenergetics and oxidative stress, plays a central role in diseases like Alzheimer’s, Parkinson’s, and cardiomyopathy. Mitochondrial dysfunction and reduced enzyme activity may further contribute to the loss of NAD+, which impairs sirtuin and PARP function, exacerbating DNA damage, metabolic derangements, and cellular senescence – and these cellular dysfunctions drive age-related decline in organ function, frailty, and disease susceptibility. It’s a domino effect that begins at the molecular level and ends with your doctor telling you things you’d rather not hear.
The Information Theory of Aging: Your Body Is Losing Its Own Data

One of the most revolutionary ideas in aging science comes from thinking of your cells not just as biological machines, but as information systems. Research builds on findings that aging is driven by chemical changes in DNA rather than irreversible damage. In a major scientific milestone, Sinclair’s team is preparing to launch human clinical trials aimed at reversing the aging process, with trials expected to test epigenetic programming therapies designed to restore cells to a more youthful state. The idea isn’t just that cells get damaged – it’s that the system for reading and executing biological instructions gets scrambled.
The key, according to this view, is activating newly discovered vitality genes, the descendants of an ancient genetic survival circuit that is both the cause of aging and the key to reversing it. Using modified Yamanaka genes, a set of genetic factors known to reprogram cells, Sinclair’s team has already demonstrated the ability to reverse aging in animal tissues by up to 75 percent within weeks. I know it sounds like science fiction, but this work was presented at a major government summit in 2026 as near-term clinical reality. That’s extraordinary.
When Does Aging Really Accelerate? It’s Not When You Think

Most people assume aging is a gradual, linear slide – a little worse every year, like a car losing value the moment it leaves the lot. But emerging science tells a very different story. A new study found that by focusing on aging-related protein changes in the body, there is an acceleration in aging of organs and tissues around the age of 50. Of these proteins, scientists found that expressions of 48 of them linked to diseases increased with age, such as cardiovascular and liver disease. Your fifties aren’t just when you start thinking about retirement – they’re when your biology quietly starts shifting gears.
Past studies show that human aging doesn’t necessarily happen at the same pace throughout our life. This has enormous practical implications. If your body undergoes a distinct acceleration of aging around the age of 50, then targeting that window with interventions could be one of the most effective strategies for extending healthy life. One of the biggest takeaways from recent research is that aging is not driven by a single biological mechanism. It’s a perfect storm of converging processes, and timing matters more than most of us ever realized.
The New Wave of Treatments: Senolytics, NAD+ Boosters, and Epigenetic Reprogramming

Let’s be real – knowing why you age only becomes truly powerful if it leads to doing something about it. The good news is that science is catching up fast. Longevity science has recently achieved a remarkable breakthrough with the development of senolytics – compounds that selectively induce death in senescent cells, thereby reducing their detrimental impact on tissues. These agents actively eradicate dysfunctional cells, rejuvenating tissues and improving physiological function, and preclinical studies demonstrate that clearing senescent cells can enhance tissue function, delay onset of age-related diseases, and even extend lifespan. Some scientists compare senolytics to a biological housecleaning service that sweeps out the cells doing the most damage.
Interventions targeting primary hallmarks of aging – such as telomerase gene therapy to counter telomere attrition, NAD+ precursors like NMN to support DNA repair and genomic stability, partial epigenetic reprogramming with Yamanaka factors to reverse transcriptional aging, and autophagy enhancers like rapamycin to restore proteostasis – have shown promise in preclinical models. Sinclair has said advances in biotechnology are rapidly improving scientists’ ability to control human biology, and he predicted that within the next 10 to 20 years, modern healthcare systems could appear outdated as treatments shift toward preventing and reversing aging itself. That’s either the most hopeful or most mind-bending sentence in modern medicine, depending on your perspective.
Conclusion: The Age of Aging Is Just Beginning

We are living through a genuinely historic moment in science. The old story – that aging is simply what happens when time passes, and there is nothing to be done – is being dismantled piece by piece. Research underscores that targeting multiple hallmarks of inflammation, metabolism, senescence, mitochondrial function, and nutrient signaling leads to better outcomes than focusing narrowly on any one pathway. In other words, aging is not one problem with one solution. It is a concert of biological failures, and the scientists working on it are learning to conduct that orchestra.
The most exciting part isn’t even the treatments themselves. It’s the shift in mindset. A major aim in aging research is to separate the unavoidable progression of chronological time from the harmful decline in function with biological age. You don’t have to accept that getting older means falling apart. Your biological age and your birth year do not have to match. That’s not a marketing slogan – in 2026, it’s an emerging scientific reality.
We’re not at the finish line yet. Huge challenges remain, including translating laboratory breakthroughs into safe, accessible treatments for real people. Still, the direction of travel is unmistakable. The next decade of aging research may well be the most consequential in human history. So here’s a question worth sitting with: if science could genuinely slow, halt, or reverse your biological aging, what would you do with all that extra healthy time? What do you think about it? Tell us in the comments.




