Every year, millions of people worldwide live with a virus quietly destroying their liver, often without even knowing it. Hepatitis C is one of medicine’s most stubborn adversaries, and despite decades of research, a working vaccine has remained frustratingly out of reach.
What scientists have recently unveiled could change that story entirely. A nanoparticle-based vaccine strategy is showing early but genuinely exciting promise, and the science behind it is far more clever than it might first sound. Let’s dive in.
Why Hepatitis C Has Been So Hard to Vaccinate Against
Here’s the thing about Hepatitis C virus, or HCV: it’s a shapeshifter. The virus mutates rapidly, meaning your immune system barely gets a chance to recognize it before it changes its appearance entirely. It’s a bit like trying to catch someone who changes their face every few hours.
Unlike hepatitis A and B, there are no licensed vaccines for hepatitis C despite the disease affecting an estimated roughly 50 million people globally. The core problem is that HCV presents so many genetic variants that a traditional vaccine targeting one strain often fails completely against another. Scientists have long searched for a vulnerability the virus cannot easily mutate away from.
The Nanoparticle Strategy: A Smarter Way to Train the Immune System
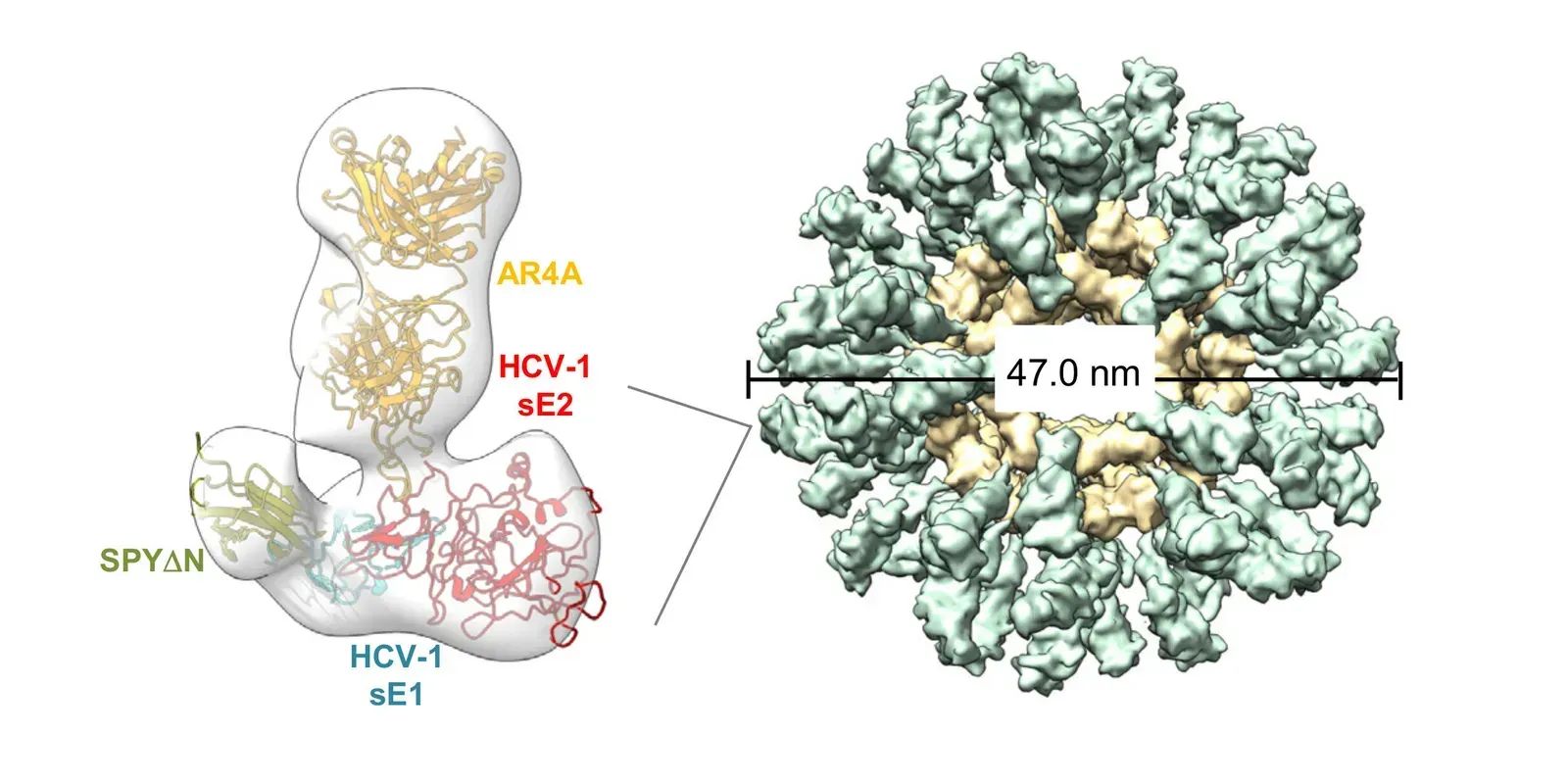
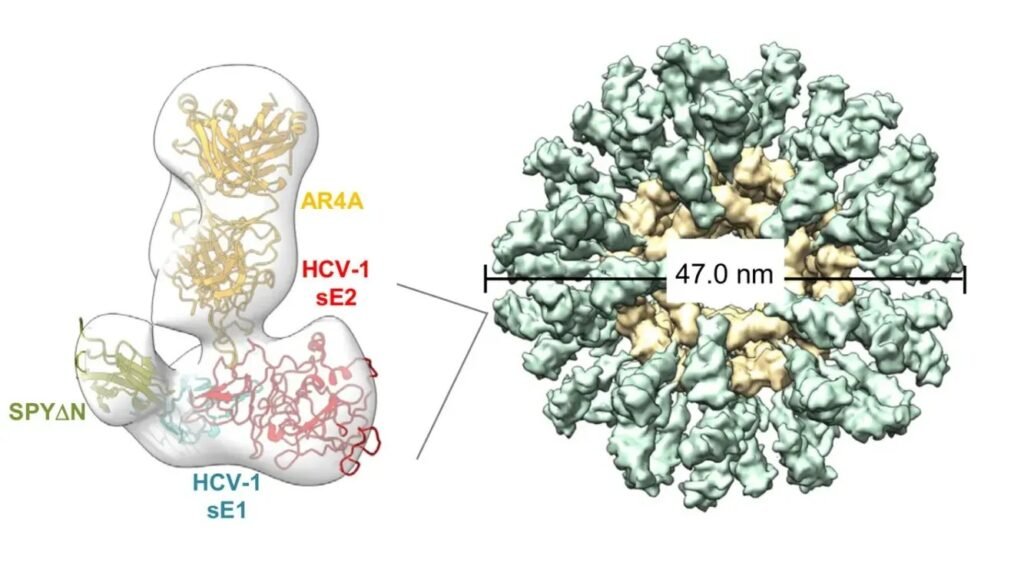
Researchers have developed a nanoparticle vaccine platform that displays multiple copies of a specific HCV protein on its surface, essentially creating a highly organized, repeated visual target for the immune system. Think of it like posting the same wanted poster on every wall in a city rather than handing out a single flyer. The immune system responds far more aggressively to repeated, structured signals.
The key protein being targeted is the E2 envelope glycoprotein, which sits on the outer shell of the hepatitis C virus and plays a critical role in how the virus latches onto liver cells. Because this protein is essential for the virus to function, HCV cannot easily discard or dramatically alter it without losing its own ability to infect. That makes it a particularly compelling target for vaccine design.
What Makes These Nanoparticles Special
The nanoparticles used in this research are self-assembling protein structures, tiny molecular scaffolds that organize themselves into precise geometric shapes. Attaching viral proteins to these scaffolds in a uniform, repeating pattern appears to dramatically amplify the immune response compared to a solitary protein presentation. Honestly, the elegance of it is kind of remarkable.
What scientists found is that the nanoparticle-displayed E2 protein triggered notably stronger antibody responses in preclinical models than the same protein presented in a conventional format. The geometry and density of antigen display seems to matter enormously to the immune system, a detail that had perhaps been underestimated in earlier vaccine design efforts.
Broadly Neutralizing Antibodies: The Holy Grail
The most exciting element of this research is the type of antibodies the vaccine appears to provoke. Broadly neutralizing antibodies, sometimes called bNAbs, are special immune weapons capable of neutralizing a wide range of viral variants rather than just one specific strain. In HIV research, the pursuit of bNAbs has dominated vaccine science for years because of their potential to overcome viral diversity.
For hepatitis C, generating broadly neutralizing antibodies would be a game-changer of almost incomprehensible significance. The nanoparticle approach in this study appears to stimulate the immune system toward producing exactly these kinds of broad-spectrum antibodies, suggesting the vaccine platform could potentially work across the diverse genetic landscape of HCV. It’s hard to say for sure how far this will translate into human trials, but the early signals are genuinely hard to dismiss.
Preclinical Results and What They Actually Mean
The research team tested their nanoparticle vaccine in animal models, observing strong immune responses and the production of antibodies that could neutralize multiple HCV genotypes. These are preliminary findings, important to say clearly, and animal results do not always translate cleanly into human outcomes. That caution is real and worth holding onto.
Still, the breadth of neutralization seen across different HCV strains in these early experiments is notable by any standard. Scientists reported that immunized animals developed antibodies targeting conserved regions of the E2 protein, meaning parts of the protein that remain relatively stable across viral variants. That consistency is exactly what you’d need in a vaccine intended to protect against a virus as genetically promiscuous as HCV.
The Broader Implications for Vaccine Science
What this research really demonstrates goes beyond hepatitis C alone. The nanoparticle platform being used here is flexible enough in principle to be adapted for other challenging viruses where antigenic diversity has long blocked vaccine development. HIV, RSV, and certain influenza strains all share this problem of rapid mutation and immune evasion.
The concept of using geometric nanoparticle scaffolds to amplify and direct immune responses is increasingly seen as one of the more powerful tools in next-generation vaccine design. If this hepatitis C application succeeds in clinical development, it could validate a broader template that researchers apply to entirely different diseases. The ripple effects of a single successful platform like this could be enormous for global public health.
What Comes Next on the Road to a Real Vaccine
Translating these findings into a human vaccine involves several more phases of testing, including safety studies, dose optimization, and eventually large-scale clinical trials. That process typically takes years, sometimes over a decade, and many promising candidates have stumbled along the way. Measured optimism, not celebration, is the right mood here.
The research team is reportedly working to refine the nanoparticle design further and optimize how the E2 protein is displayed to maximize the broadly neutralizing antibody response. The goal moving forward is to build a candidate strong and consistent enough to warrant progression into early human trials. A licensed hepatitis C vaccine would represent one of the most significant infectious disease breakthroughs in recent memory, given how long this particular challenge has resisted solution.
Conclusion
Let’s be real: the world has been waiting for a hepatitis C vaccine for a very long time. Tens of millions of people are living with a preventable, liver-damaging infection, and the tools to stop new transmissions have been woefully incomplete. The fact that antiviral treatments for HCV now exist is wonderful, but treatment is not the same as prevention.
I think this nanoparticle research deserves serious attention precisely because it attacks the core biological problem rather than trying to work around it. The strategy of targeting a conserved, functionally essential region of the virus and amplifying the immune response through geometric protein display is not a workaround. It’s a direct assault on HCV’s greatest strength, its genetic versatility. Whether this particular candidate clears every hurdle ahead remains to be seen, but the science here feels genuinely different from what came before.
What do you think: could a hepatitis C vaccine finally be within reach, or has this virus surprised us too many times before? Tell us in the comments.