Walk into almost any cutting-edge biology lab in 2026 and you can feel it: a quiet, electric confidence that human aging is finally giving up some of its best-kept secrets. Not in the flashy sense of a single “immortality gene,” but in a slower, more surprising way, as patterns emerge from massive genomic datasets, long-term health records, and experiments that rewind the cellular clock. The new story of longevity is not about escaping death, but about stretching the healthy years we have, pushing back frailty, and sidestepping the diseases that most often cut lives short. What is changing now is that we can watch, in unprecedented detail, how our DNA and its regulation shape those trajectories over decades. This article looks at a few of the most powerful genetic discoveries reshaping our understanding of health and lifespan – and what they might realistically mean for any of us hoping to grow very old and still feel very alive.
From Single “Longevity Genes” To Polygenic Maps Of Long Life
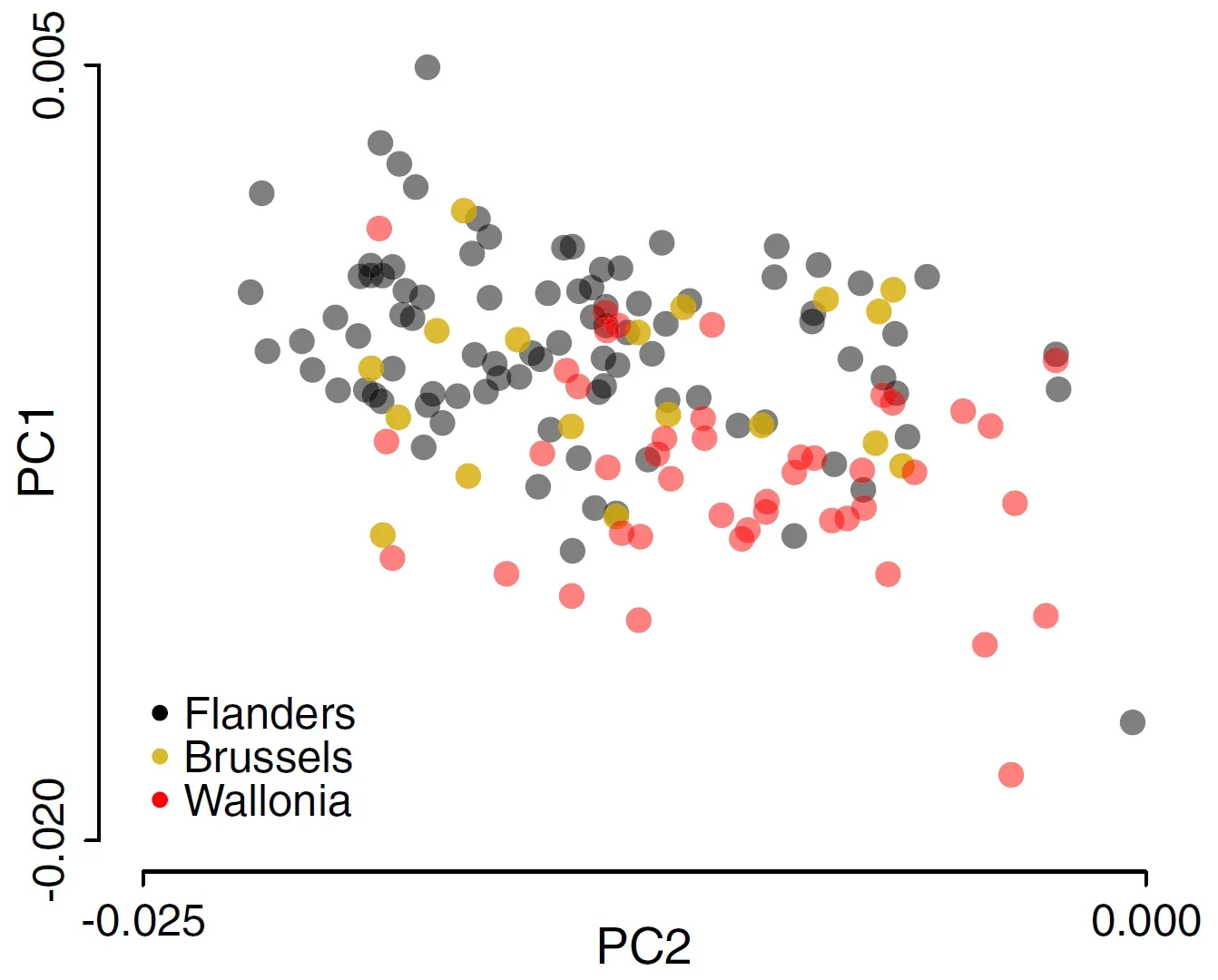
Not long ago, the hunt for longevity seemed to revolve around a handful of celebrity genes: APOE, FOXO3, maybe a few others that showed up again and again in centenarians. Those signals are still important, but large human studies have made it clear that lifespan is influenced by thousands of tiny genetic nudges, each shifting risk by a small amount. Researchers now build polygenic risk scores that combine these nudges into a single estimate of a person’s genetic tilt toward longer or shorter life, or toward specific age-related diseases.
These scores are already being linked to everything from heart disease and type 2 diabetes to resilience against cognitive decline. The surprising twist is that genetic variants associated with longer life almost always act by lowering the risk of common killers, rather than by slowing some abstract “aging clock.” That means the promise of polygenic tools is not predicting exactly when we die, but identifying which pathways – cholesterol handling, inflammation, blood pressure control – we can target to keep people healthy for longer, regardless of their starting DNA hand.
APOE, Alzheimer’s, And The Genetics Of Keeping The Brain Young
Few genetic discoveries have changed the conversation about aging brains as dramatically as APOE, the gene that strongly shapes risk for late-onset Alzheimer’s disease. One version of the gene, often called E4, greatly increases the chance of developing the disease, while another, E2, appears to protect some people into extreme old age. For years this felt like a cruel genetic lottery: either you drew the risk variant or you did not.
What has shifted in the last several years is a more nuanced view of how APOE interacts with lifestyle, other genes, and potential treatments. Brain imaging and fluid biomarkers now show that people with the high-risk variant can sometimes keep pathology at bay for longer if they aggressively manage cardiovascular health, sleep, and metabolic factors. At the same time, experimental therapies are aiming upstream, testing antibodies and RNA-based approaches that modulate APOE and related pathways directly. The dream is not to erase risk, but to turn a genetic disadvantage into a manageable factor, the way blood pressure drugs once transformed the risk landscape for stroke.
Decoding Centenarians: What Exceptionally Long-Lived Families Reveal

Families in which living past ninety is almost normal have become a kind of natural experiment for geneticists. When researchers sequence the genomes of centenarians and their relatives, they often find clusters of rare variants that tweak immune responses, lipid metabolism, and insulin signaling in subtle ways. No single mutation grants extreme longevity, but the combined effect of many small protections appears to delay multiple diseases at once.
What stands out about these individuals is not that they avoid all illness, but that they compress most serious illness into a relatively short period at the very end of life. Genetic studies suggest that they often carry fewer harmful variants overall, especially in pathways tied to DNA repair and cellular stress responses. By cataloging these protective patterns, scientists hope to design drugs or gene-based interventions that mimic the “longevity profiles” seen in these families. The ethical and technical hurdles are significant, but the core idea is simple: learn from outliers whose biology is already doing what we want medicine to do.
Epigenetic Clocks: Reading Biological Age In Chemical Marks
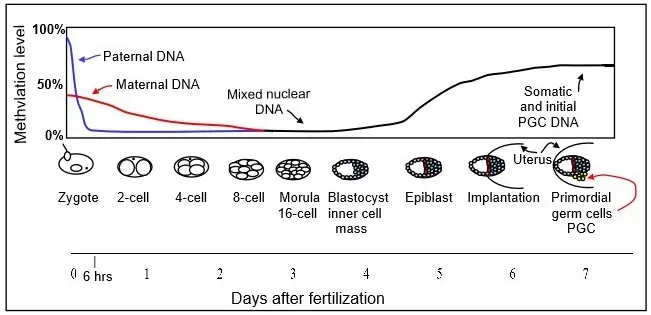
One of the most striking genetic-adjacent breakthroughs is not about changing DNA at all, but about reading its chemical decorations. Epigenetic clocks use patterns of DNA methylation – tiny chemical tags on the genome – to estimate a person’s biological age more accurately than their birth certificate can. When these clocks show a higher biological age than chronological age, people tend to have higher risks of chronic disease and earlier mortality.
This has turned epigenetic age into a kind of running scorecard for longevity research, including in trials of lifestyle changes and experimental therapies. Some small human studies report that targeted interventions – combining diet, exercise, sleep optimization, and sometimes drugs – can slow or even modestly reverse epigenetic aging markers over a couple of years. Scientists still debate whether these changes truly reflect slowing of underlying aging or just shifts in specific tissues and pathways. But for the first time, researchers can test claims about longevity with something more precise than waiting decades to count who dies when, and that is rewriting how this field operates.
Gene Editing Meets Aging Biology: Precise Tools, Messy Questions
The arrival of CRISPR-based gene editing raised an obvious and provocative question: if we can edit disease-causing mutations, could we eventually edit pathways that accelerate aging itself? In animal models, researchers have already used gene editing to tweak growth-related hormones, lipid regulators, and DNA repair machinery, sometimes producing mice that live significantly longer and stay healthier late in life. These experiments illuminate which pathways are most powerful for controlling aging at the cellular level.
In humans, however, the bar is far higher, both scientifically and ethically. Most age-related traits are shaped by many genes, so editing one target may have trade-offs we do not yet see, especially over decades. There is also a stark difference between correcting a severe inherited disorder in a child and editing relatively healthy adults in the hope of gaining extra years. For now, gene-editing work in longevity focuses mainly on diseases that strongly shorten life, such as certain cardiovascular or lipid disorders, where benefits are clearer. Still, the conceptual shift is enormous: aging is no longer an untouchable background process, but a network of levers that, at least in principle, can be moved.
Longevity Drugs From Genetic Clues: From PCSK9 To Senolytics

Some of the most practical longevity advances so far have come from translating genetic insights into drugs that mimic nature’s protective variants. A classic example is PCSK9: people born with naturally low-functioning versions of this gene tend to have very low LDL cholesterol and far fewer heart attacks across their lives. That discovery helped drive the development of PCSK9 inhibitor drugs, which now dramatically lower cholesterol and may reduce cardiovascular events beyond what traditional statins can achieve.
At the same time, genetic and transcriptomic studies of aging cells have fueled the rise of senolytics, drugs that target senescent cells – damaged cells that stop dividing but refuse to die, secreting inflammatory signals. In mice, clearing these cells can improve physical function and extend healthy lifespan, and early human trials are now testing whether similar benefits appear in specific diseases like lung fibrosis or kidney disorders. These therapies are still in their early days, and side effects remain a concern because senescent cells also play roles in wound healing and tumor suppression. Yet the logic is compelling: if age-related diseases share common cellular culprits, drugs that target those culprits might push back multiple conditions at once, effectively stretching the healthy middle of life rather than just the tail end.
Rethinking Lifestyle Advice Through A Genetic Lens
There is a quiet but important shift happening in how doctors and researchers talk about lifestyle and genes. For years, the message was almost moralistic: live right and you will age well, live badly and you will not. Large genetic studies complicated that picture, showing that some people with high genetic risk for conditions like heart disease can nearly cancel out much of that risk through aggressive lifestyle management, while others with low genetic risk can squander their advantage through smoking, poor diet, and inactivity.
What genetics adds is nuance rather than fatalism. A person with a strong inherited risk for early heart attacks might be counseled to start cholesterol-lowering strategies much earlier than guidelines based only on age or short-term risk. Another person might learn that they metabolize certain nutrients differently and respond better to specific dietary patterns. This is not an excuse to outsource responsibility to DNA; if anything, it raises the stakes of knowing your own risk profile. The emerging idea is that the best longevity plan is intensely personal, shaped by genomic data but executed through everyday habits.
Why These Discoveries Matter: Aging As A Treatable Biological Process

Step back from the individual findings and a bigger pattern emerges: aging is starting to look less like an inevitable slide and more like a set of partially modifiable processes. Genetic discoveries have linked longer life not to a mystical fountain of youth, but to improved maintenance of systems we already understand – vascular health, immune balance, metabolic flexibility, and the capacity to repair damage. That may sound less glamorous, but it is far more actionable, both for medicine and for individual choices.
Compared with older views that treated aging as a black box or purely as wear and tear, this new framework is radically different. It suggests that the same tools we use to tackle cancer or heart disease – careful clinical trials, targeted drugs, preventative screening – can also be pointed at the underlying biology that makes those diseases so common with age. At the cultural level, this challenges the idea that adding years automatically means adding suffering and dependency. The scientific goal is not simply to extend lifespan, but to extend healthspan, so that more of us reach old age with minds and bodies that still feel usable, even joyful. That shift in emphasis might be the most important discovery of all.
Where Longevity Genetics Goes Next – and What You Can Do Now
The next decade of longevity genetics will likely be defined less by spectacular single-gene discoveries and more by scale and integration. Massive biobanks are combining genomic data with decades of medical records, imaging, and lifestyle information, allowing researchers to trace how specific variants play out over whole lives. New tools will likely make genetic risk scores more accurate across diverse ancestries, addressing a major current bias toward European-ancestry data. At the same time, there will be growing pressure to translate these insights into interventions that are affordable and equitably available, not just boutique services for the wealthy.
For individuals, the most realistic path to benefiting from all this science is surprisingly grounded. Talk with your doctor about family history and, when appropriate, genetic testing for clearly actionable risks, like certain cholesterol disorders or cancer syndromes. Pay attention to clinical trials that focus on prevention and early intervention, not just rescue therapies after disease has fully taken hold. Support policies and research initiatives that prioritize long, healthy lives as a public good rather than a luxury. In the end, the most powerful message of modern genetics is not that your fate is written in code, but that understanding that code can help you make smarter choices about the years you have. What will you do with that knowledge?

Suhail Ahmed is a passionate digital professional and nature enthusiast with over 8 years of experience in content strategy, SEO, web development, and digital operations. Alongside his freelance journey, Suhail actively contributes to nature and wildlife platforms like Discover Wildlife, where he channels his curiosity for the planet into engaging, educational storytelling.
With a strong background in managing digital ecosystems — from ecommerce stores and WordPress websites to social media and automation — Suhail merges technical precision with creative insight. His content reflects a rare balance: SEO-friendly yet deeply human, data-informed yet emotionally resonant.
Driven by a love for discovery and storytelling, Suhail believes in using digital platforms to amplify causes that matter — especially those protecting Earth’s biodiversity and inspiring sustainable living. Whether he’s managing online projects or crafting wildlife content, his goal remains the same: to inform, inspire, and leave a positive digital footprint.




