Imagine waking up one day to discover that aging has been reversed. Your skin regains its elasticity, your energy levels soar, and your body feels decades younger than it did yesterday. Sounds like pure science fiction, right? Yet research happening right now in laboratories around the world suggests this might not be as far-fetched as you think.
The quest to unlock the secrets of aging has captivated scientists, billionaires, and everyday people alike. From tiny cellular structures called telomeres to experimental drugs being tested in clinical trials, humanity stands at an intriguing crossroads. The question is no longer just about adding years to life, but adding life to years. So let’s dive into what science has uncovered about turning back our biological clocks.
Understanding the Biological Clock Within Our Cells

Cellular senescence, a hallmark of aging defined by stable exit from the cell cycle in response to cellular damage, has been directly implicated as a key driver of aging and age-related diseases. Think of senescent cells as retired workers who refuse to leave the office. They stop dividing but stick around, creating chaos by releasing inflammatory molecules that damage nearby healthy cells.
Recent experimental evidence has shown that the genetic or pharmacological ablation of senescent cells extends lifespan and improves healthspan. Researchers have discovered that when you clear these troublemakers from tissues, remarkable things happen. Mice in experiments live longer and healthier lives. However, here’s the thing: senescence isn’t entirely villainous.
Senescence is actually a protective strategy that cells use when faced with stress or damage, and it’s also needed for tissue regeneration and development. It helps with wound healing and prevents damaged cells from becoming cancerous. The complexity lies in finding the sweet spot between keeping beneficial senescence while eliminating the harmful accumulation of senescent cells that comes with aging.
Telomeres: The Ticking Time Bombs at Chromosome Ends
Telomere shortening is a well-known hallmark of both cellular senescence and organismal aging, and telomere length has been recognized as one of the best biomarkers of aging. Imagine telomeres as the plastic tips on shoelaces. Each time your cells divide, these protective caps get shorter, and when they become critically short, the cell can no longer function properly.
Daily vitamin D3 supplementation was found to prevent the shortening of telomeres, the protective ends on chromosome strands, which is a hallmark of aging. This discovery from Harvard researchers in 2025 represents a fascinating breakthrough. Something as simple as taking a vitamin supplement might help preserve these crucial structures.
As a normal cellular process, a small portion of telomeric DNA is lost with each cell division, and when telomere length reaches a critical limit, the cell undergoes senescence or apoptosis. Lifestyle factors play a massive role here. Smoking, poor diet, and chronic stress can accelerate telomere shortening, while exercise, good nutrition, and stress management may help preserve them.
Senolytics: The Drugs That Target Zombie Cells
Senotherapeutics have been developed to pharmacologically eliminate senescent cells, termed senolytics, or suppress their harmful effects, termed senomorphics. These drugs represent one of the most exciting frontiers in anti-aging research. The concept is beautifully simple: if senescent cells cause aging problems, why not just remove them?
A 2025 preclinical study showed that the senolytic combination of dasatinib and quercetin reduced senescent cell burden, inflammatory signaling, and bone loss in aging periodontal tissues. The combination pairs a cancer drug with a plant compound found in foods like onions and apples. Early results are promising, though researchers emphasize caution about completely eliminating senescence.
Senescence is context dependent – it can be harmful, but it can also be necessary. This complexity means that future senolytic therapies will need to be highly targeted and carefully timed. The goal isn’t to eliminate senescence altogether but to clear out the excessive accumulation that occurs with age.
The NAD+ Molecule: A Master Regulator of Aging
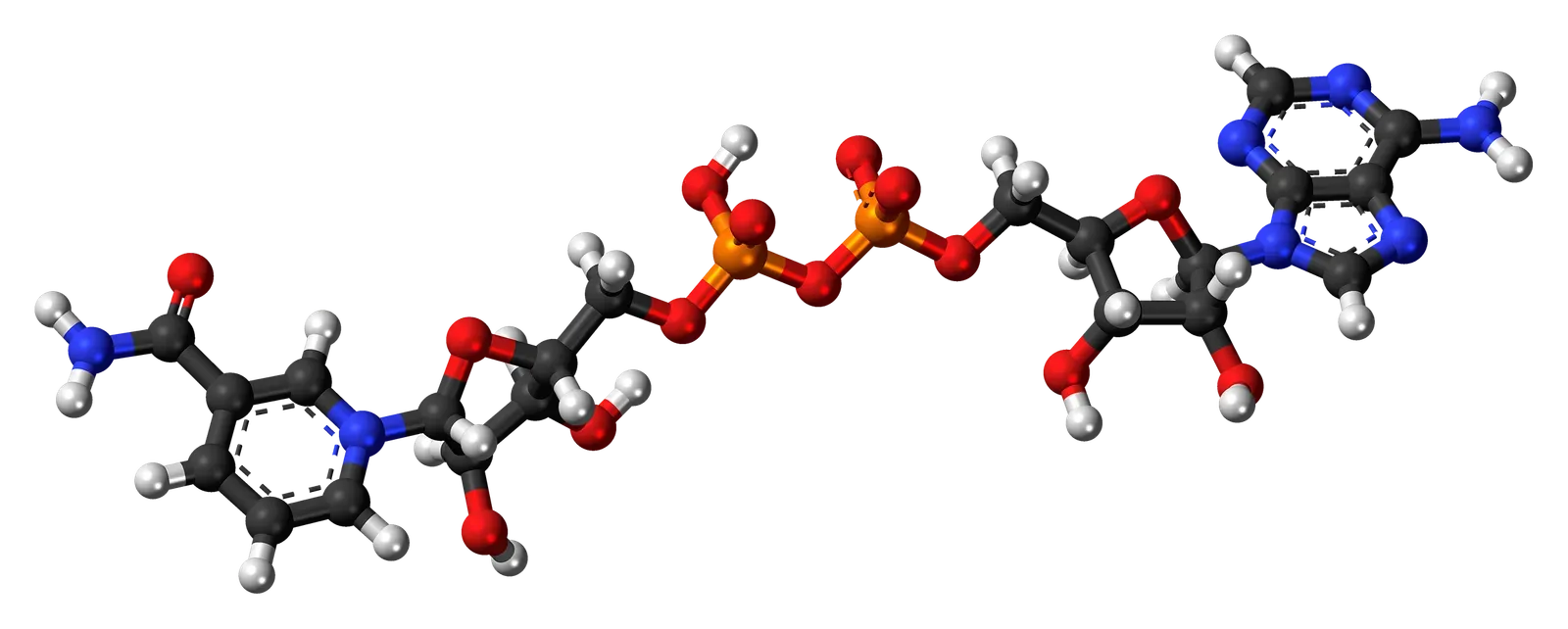
NAD+ is a coenzyme that is essential for metabolism, but natural production of that coenzyme does decline as you age, and supplementing NAD+ precursors can help increase production of the molecule in the body. This molecule sits at the center of countless cellular processes, from energy production to DNA repair. Without adequate NAD+, cells simply can’t function optimally.
In mice, successful restoration of NAD+ to youthful levels resulted in cardiovascular improvements, reversal of multiple metabolic conditions, improvements to muscle function and endurance, and increased mitochondrial function. The animal research sounds almost too good to be true. Scientists observed improved cognition, enhanced organ protection, and even better fertility in models where NAD+ levels were boosted.
The clinical evidence that raising NAD+ concentrations can improve physiological function is unclear, and most trials performed to date have consisted of relatively small sample sizes. Let’s be real: the human data isn’t nearly as compelling yet. While supplements containing NAD+ precursors like NR and NMN can raise NAD+ levels in blood, whether this translates to actual health benefits remains an open question requiring much larger studies.
Rapamycin: From Organ Transplants to Longevity Labs
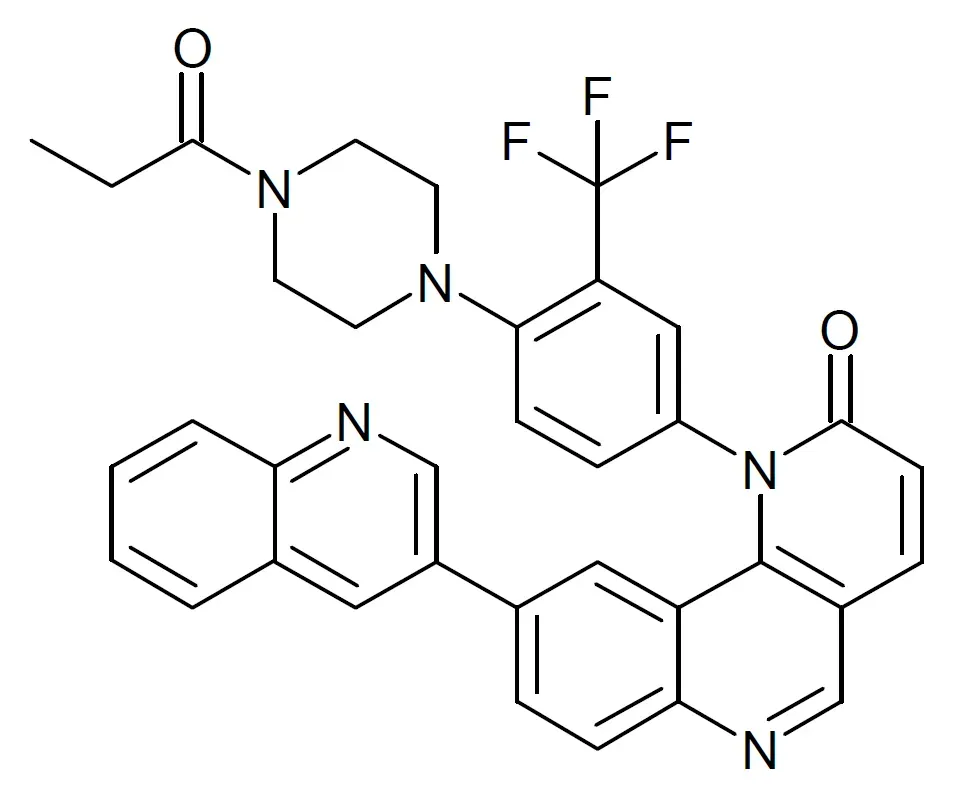
A 2025 pilot clinical study investigated whether short-term inhibition of mTOR with low-dose rapamycin could improve cardiovascular function in six healthy male participants aged 70 to 76 who took 1 mg daily for 8 weeks, revealing statistically significant improvements in diastolic heart function. Rapamycin’s journey from an immunosuppressant drug to a potential fountain of youth compound is remarkable.
The recently published PEARL trial demonstrated that low-dose intermittent rapamycin was well tolerated over 1 year and resulted in modest changes in biomarkers of biological aging. This represents the first major human trial specifically targeting aging itself. The fact that participants tolerated the drug well is encouraging, though honestly, the benefits were modest rather than dramatic.
Results published showed median and maximum lifespan extension in both sexes, which was remarkable, especially such a response in older mice that were expected to die from treatment. The mouse data continues to be extraordinarily promising. When elderly mice received rapamycin, they lived longer and healthier lives, defying initial concerns that the treatment would be too harsh for aged animals.
Cellular Reprogramming: Turning Back the Cellular Clock
In 2020, a study showed that gene therapy activating genes coding for Yamanaka factors can reverse signs of aging, such as vision loss, in mice, illustrating that activating these factors could help rejuvenate tissues throughout the body. This breakthrough represents one of the most ambitious approaches to reversing aging. Yamanaka factors are proteins that can essentially reset cells to a more youthful state.
Harvard’s Dr. David Sinclair predicts that pills will be available within the next 10 years that target certain genes to reverse aging in tissues throughout the body, and believes that one day scientists may find a way to double the human lifespan. It’s hard to say for sure whether these predictions will come true, but the underlying science is undeniably fascinating.
Life Biosciences announced that its Partial Epigenetic Reprogramming therapy, ER-100, is in the works to begin the first-ever human clinical trials in early 2026, and preclinical data showed therapeutic impact across systems. We’re moving from mice to humans faster than many expected. The next few years will reveal whether cellular reprogramming can deliver on its extraordinary promise.
Mitochondria: The Powerhouses Holding Anti-Aging Secrets
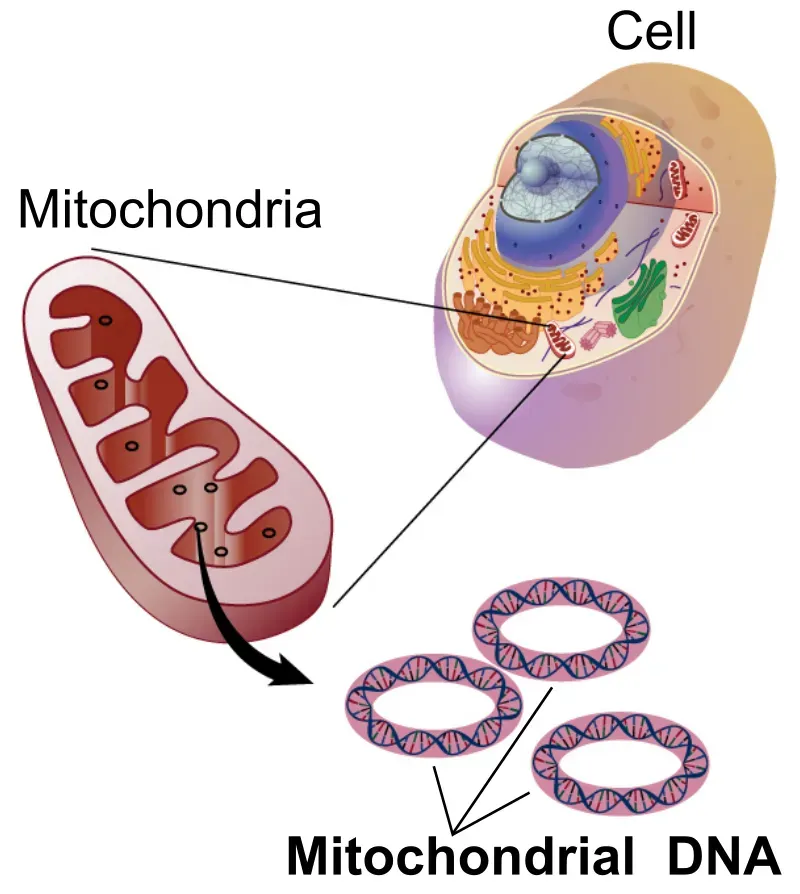
Mitochondria, often called the powerhouse of the cell, generate the energy that keeps cells running, and because aging and many age-related diseases are closely tied to declining mitochondrial function, scientists see mitochondria as an important target. When these cellular power plants falter, everything else follows. Energy production drops, oxidative stress increases, and cells age faster.
On average, mice engineered to produce higher levels of the protein COX7RP lived 6.6 percent longer than wild-type mice, and the benefits were not limited to lifespan alone, since the engineered mice also showed signs of better healthspan. This 2025 discovery points to a potential new target for slowing aging at the cellular level.
Stimulating lysosome activity can restore cellular cleanup function, helping remove harmful proteins and reducing signs of cell aging, and by reactivating this system, scientists were able to help cells recover their youthful behavior. The body already has built-in recycling systems designed to clear cellular garbage. Finding ways to reactivate these systems as we age could provide a more natural path to longevity.
Lifestyle Interventions: The Proven Path to Healthier Aging

An April 2025 study found that transcendental meditation can significantly alleviate stress and slow down aging, with participants showing lower expression of genes associated with inflammation and aging. Sometimes the most powerful interventions don’t come in pill form. Meditation’s ability to reduce biological aging demonstrates the profound connection between mind and body.
Research published in May 2025 found that supplementing with daily vitamin D3 can reduce biological wear and tear equivalent to nearly three years of aging. Simple, inexpensive interventions like vitamin D supplementation might offer meaningful benefits. This kind of accessible approach democratizes longevity science beyond expensive treatments.
A study found that social relationships can slow cellular aging. Here’s something that might surprise you: your friendships could be as important as any supplement. Strong social connections appear to influence aging at the cellular level, reminding us that longevity isn’t just about biology but also about how we live our lives.
Where Do We Stand on Living Forever?

Speaking at Bank of America’s Breakthrough Technology Dialogue in February 2025, Dr. Zhavoronkov predicted that it will not only become commonplace to live to 120 but to live well to 120. The shift from lifespan to healthspan represents a crucial evolution in thinking. Nobody wants to simply exist for 120 years; people want those years to be vibrant and fulfilling.
2025 marked one of the most productive years in human longevity science, with major breakthroughs in human trials, drug repurposing, metabolic therapeutics, and landmark publications that reshaped thinking on rapamycin, GLP-1, and other compounds. The pace of progress is genuinely accelerating. What seemed like distant possibilities a decade ago are now entering clinical trials.
We have early evidence that certain strategies do provoke rejuvenation at the cellular level in humans, and these proof-of-concept successes validate that human aging is modifiable to a degree. The evidence is mounting, though we need to stay grounded. Modest improvements in biomarkers don’t necessarily translate to dramatically extended lifespans. The journey from laboratory success to clinical reality is long and complex, filled with unexpected challenges and occasional setbacks.
The science of anti-aging has moved from fringe speculation to serious medical research. We’re uncovering the mechanisms that drive aging and developing tools to intervene in those processes. Living forever might still be science fiction, but living healthier for much longer is rapidly becoming science fact. The real question isn’t whether we can extend human healthspan – the evidence suggests we can – but how quickly we can translate these discoveries into safe, effective treatments that benefit everyone. What aspects of aging would you most want science to address? The answers might arrive sooner than you expect.

Hi, I’m Andrew, and I come from India. Experienced content specialist with a passion for writing. My forte includes health and wellness, Travel, Animals, and Nature. A nature nomad, I am obsessed with mountains and love high-altitude trekking. I have been on several Himalayan treks in India including the Everest Base Camp in Nepal, a profound experience.



