Imagine waking up one day and realizing that, biologically, you are getting younger instead of older. It sounds like science fiction, the kind of idea you’d expect in a futuristic movie, not in a medical journal. Yet over the last decade, serious scientists in real labs have started to do things to cells, mice, and even humans that look suspiciously like turning back the biological clock. The race to slow, halt, or even reverse aging is no longer fringe – it’s quietly becoming one of the most well-funded and fiercely competitive areas in modern science.
At the same time, the hype can be ridiculous, and the snake oil is very much alive and well. I’ve watched friends throw money at magic supplements and biohacking gadgets promising decades of extra life, only to feel mostly…normal. Underneath all the noise, though, there are a few breakthroughs that truly matter, backed by data rather than wishful thinking. That gap between wild marketing and quiet, careful science is where the real story of longevity is unfolding right now.
The Surprising Idea: Aging as a Treatable Condition

For most of human history, aging was just seen as inevitable decay, like rust on metal or cracks in old paint. Today a growing number of researchers treat it more like a disease process – complex, multifactorial, but in principle, something you can slow down or modify. That shift sounds philosophical, but it has very practical consequences: if aging is a process, then every step in that process is a potential target for a drug, therapy, or lifestyle intervention.
What shocks many people is that some age-related damage seems to be at least partly reversible in lab models. Old cells can be coaxed back into behaving more like young cells. Aged animals can regain endurance, cognitive sharpness, and organ function after certain treatments. We’re not talking about immortality or some fantasy of staying twenty forever; we’re talking about extending the number of years you’re healthy enough to live the way you actually want to live. If aging can be treated, then the question quietly changes from “if” to “how far can we safely go?”
Senolytics: Killing Zombie Cells to Revive Old Tissues
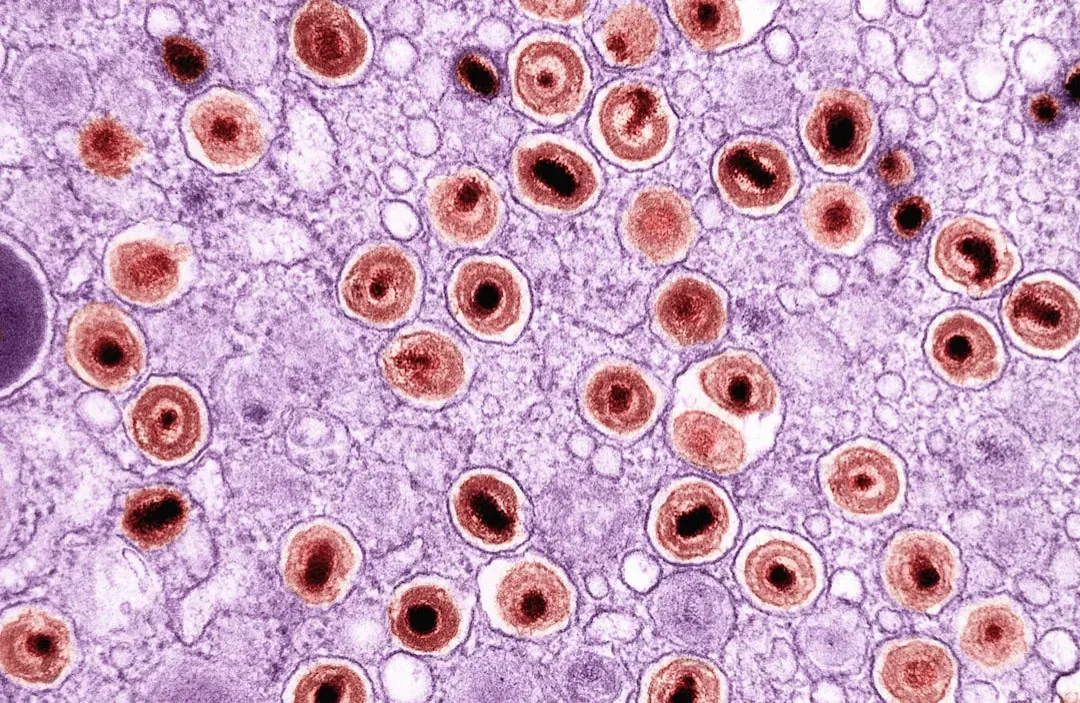
One of the most dramatic ideas in longevity research centers on so‑called “zombie cells,” known scientifically as senescent cells. These are damaged cells that stop dividing but refuse to die, hanging around like bad tenants who never pay rent and trash the place. They pump out inflammatory molecules that can disrupt nearby healthy cells and are linked to many hallmarks of aging, from joint degeneration to cardiovascular problems. As we get older, these zombie cells build up throughout the body.
Senolytic drugs are designed to seek out and destroy these senescent cells, helping tissues function more like they did in youth. In mouse studies, periodically clearing these cells has extended lifespan and, more importantly, improved healthspan – mobility, organ function, resilience to disease. Early human trials using combinations like dasatinib and quercetin in specific age-related conditions have shown promising signs, like improvements in physical function and reductions in certain inflammatory markers. It’s not a magic pill yet, and safety is a big concern, but the core idea – that strategically getting rid of harmful cells can make the whole system act younger – is one of the most thrilling shifts in how we think about aging.
Resetting the Clock: Epigenetic Reprogramming and Biological Age

If your DNA is the book of you, your epigenome is the system of bookmarks, highlights, and dog‑eared pages that tell your cells which parts of that book to read. Over time, that epigenetic pattern gets messy, and this “epigenetic drift” is strongly tied to aging. Researchers can now estimate your “biological age” from these epigenetic markers, and in many people it doesn’t match their calendar age. That alone is wild: two forty‑year‑olds can be biologically as different as a thirty‑year‑old and a fifty‑year‑old.
The next leap is even more startling: partial epigenetic reprogramming, where certain factors (inspired by the genes used to create stem cells) are turned on just enough to push cells into a more youthful state without erasing their identity completely. In animal studies, doing this carefully has restored vision in old mice and reversed some markers of aging in multiple tissues. A few early-stage human efforts are now exploring related strategies, though we’re still very much at the cautious, experimental stage. The risk of pushing cells too far – into cancer, for example – is not hypothetical. Still, the idea that we might be able to “polish” the epigenetic marks of age and dial biological age backward, even by a small amount, feels like something out of a sci‑fi novel that suddenly has footnotes and lab protocols.
Rejuvenating the Body’s Power Plants: Mitochondria and Cellular Energy
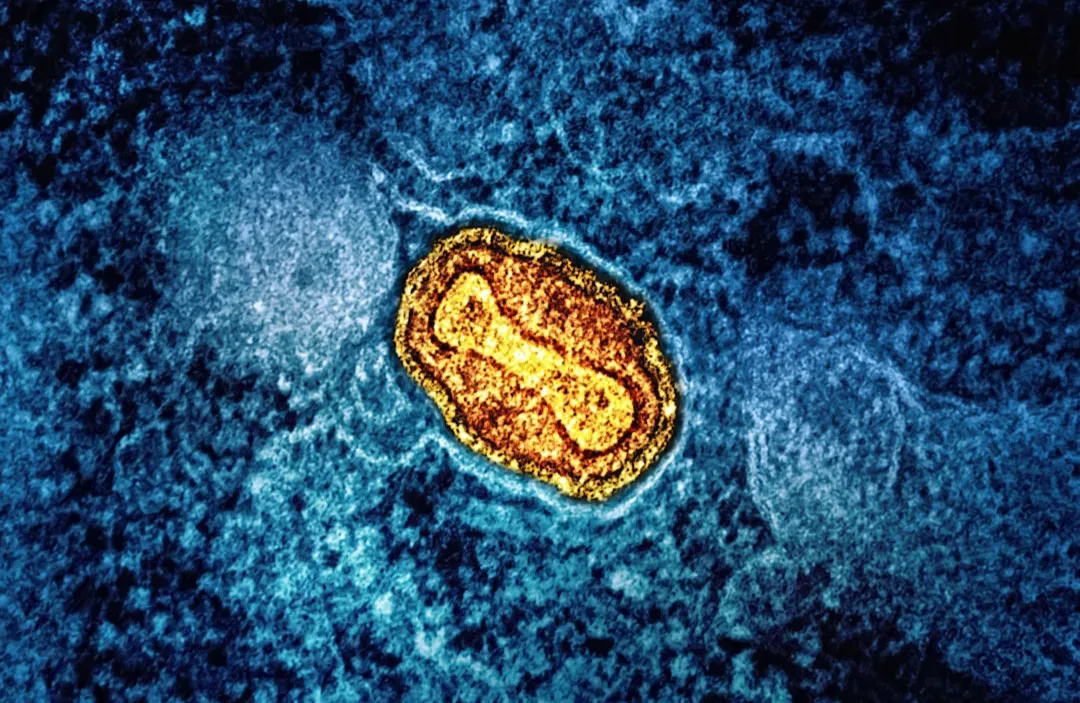
Every cell in your body runs on tiny power plants called mitochondria, and like any machinery, they wear down over time. As mitochondrial function declines, cells produce less energy and more reactive byproducts that damage proteins, fats, and DNA. Many people feel this not as something abstract, but as the creeping fatigue, slower recovery, and weaker muscles that seem to show up out of nowhere in midlife. In a very real sense, aging can feel like living with a city‑wide brownout in your cells.
Longevity researchers are trying to repair or bypass these failing power plants. Some compounds aim to boost molecules like NAD, which play a key role in mitochondrial health and cellular repair, while others try to trigger mitophagy, the process by which cells clear out defective mitochondria. Animal studies have shown that enhancing mitochondrial quality control can improve endurance, metabolic health, and even lifespan. Human trials are more mixed so far – some people report better energy and metabolic markers, others feel little change – but this is exactly the kind of area where incremental gains could add up. You might never “feel” your mitochondria getting younger, but if your heart, brain, and muscles behave like they’re from someone a decade younger, that’s a kind of everyday magic.
Blood Factors, Plasma Exchange, and the Young–Old Connection
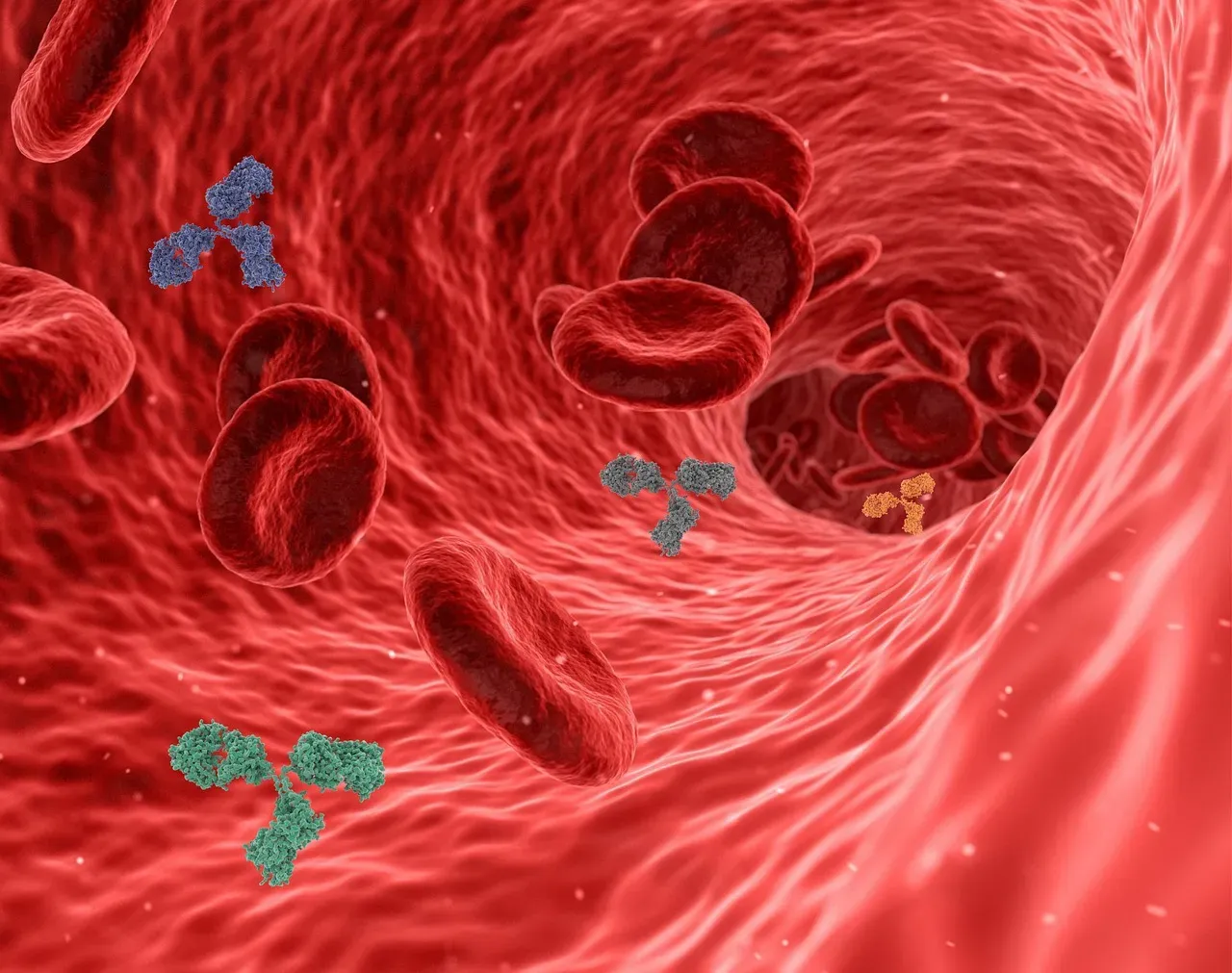
Few findings in aging research have been as eerie and captivating as studies where the circulatory systems of young and old mice are joined. In those experiments, old mice exposed to young blood show improvements in tissue repair and certain cognitive functions, while the young mice receiving old blood tend to decline faster. It sounds like a dark fairy tale, but the data forced scientists to consider that circulating factors in the blood might strongly influence how quickly tissues age or regenerate.
Modern work has moved away from the crude idea of “young blood” toward more precise approaches like plasma exchange, where components of the blood are filtered or replaced, and the hunt for specific pro‑aging and anti‑aging proteins. Early human trials of plasma dilution – removing part of a person’s plasma and replacing it with a simple solution – have hinted at improvements in inflammatory markers and some functional measures in older adults. This is far from proven rejuvenation, and there’s plenty of deserved skepticism, especially given some opportunistic clinics that jumped ahead of the evidence. Still, if we can identify and modulate the key molecules circulating in blood that push tissues toward aging or repair, that could be one of the most direct ways to nudge the entire body in a younger direction.
Gene Therapies and Designer Longevity: Editing the Rules of Aging
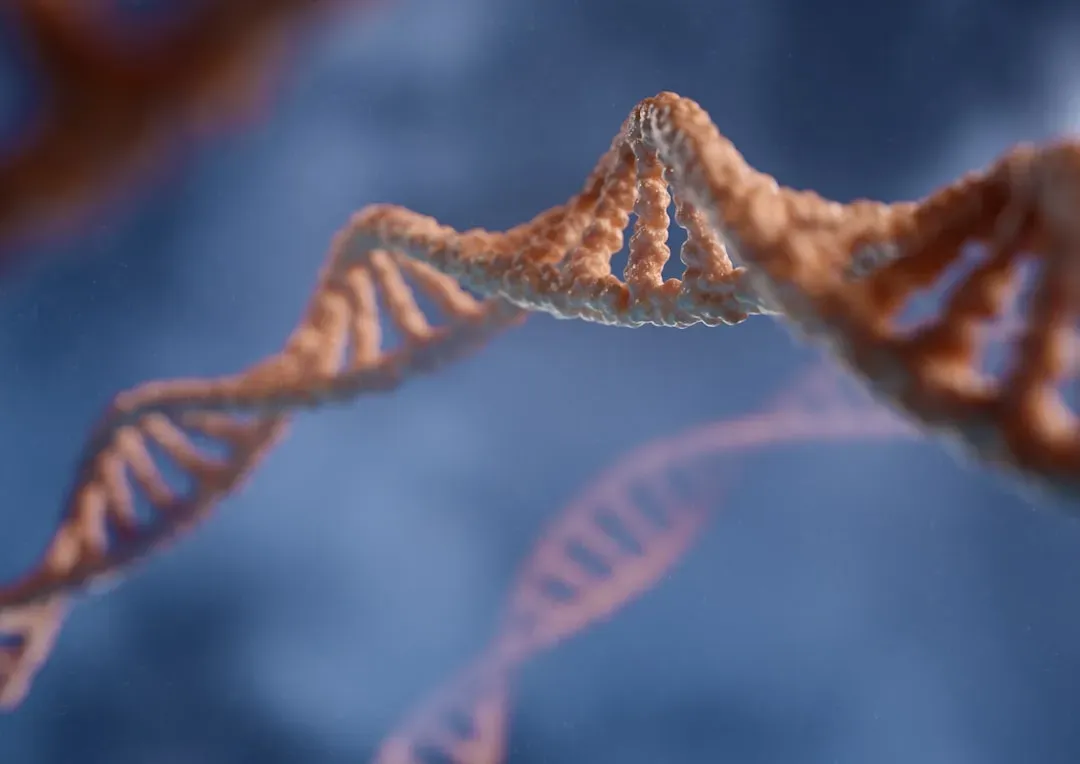
Gene therapy used to be the stuff of whispered speculation; now it’s an approved treatment for several rare diseases and moving fast into more common conditions. That same toolkit is being tested, cautiously, against aging itself. Instead of just patching one disease, the goal is to adjust or enhance pathways that influence how quickly many age-related problems develop. For example, some researchers are exploring ways to boost genes involved in repair or stress resistance, while others are investigating how to dial down genes that accelerate inflammation or tissue damage.
In animal models, gene therapies aimed at longevity pathways have extended healthy lifespan, delayed tumors, and preserved organ function. A handful of high-profile experiments have even tested gene therapies in humans for effects like increased muscle mass or improved heart function in older adults, though these are still experimental and tightly regulated. The big ethical question is obvious: if we can tweak the rules of aging, who gets access, and how do we avoid unintended consequences that might show up years later? Editing the code of life to slow aging is a lot like rewiring an airplane mid‑flight – possible in theory, massively powerful, and very unforgiving if you get it wrong.
What You Can Actually Do Now: Lifestyle as the First Longevity Drug

With all the cutting‑edge science, it’s tempting to wait for the miracle injection and ignore the boring stuff. But when you step back from the headlines and look at the most robust data, an unromantic truth jumps out: the things we already know work – sleep, nutrition, movement, stress management, social connection – still beat any single pill we have for extending healthy years. Large, long‑term studies consistently find that people who combine several healthy habits live many extra years free of major disease compared to those who do not. It’s not glamorous, but it is brutally consistent.
What’s changing now is that longevity science is finally explaining why those habits matter so much, all the way down to the level of epigenetics, mitochondria, and inflammatory pathways. Regular physical activity can act like a senolytic in some tissues, triggering the clearance of damaged cells; time‑restricted eating can influence cellular repair cycles; deep social ties reduce chronic stress hormones that accelerate biological aging. When I finally took this seriously myself – focusing on sleep and daily walking instead of compulsively chasing the latest supplement – I was annoyed to admit how much better I felt. Until the more radical therapies are proven, the most realistic form of age reversal may be closing the gap between how old you are on paper and how old your body thinks you are.
Are We Really Close to Reversing Aging?

Looking across the field, it’s clear that we can already nudge aspects of biological aging in animals and, in a few narrow ways, in humans. We can clear senescent cells, tweak epigenetic marks, improve mitochondrial quality, and manipulate blood factors, all with measurable effects on health and function. That alone is a tremendous shift from a world where aging was a black box you simply had to accept. The dream of a comprehensive, safe, and reliable age‑reversal treatment for humans, though, is still ahead of us, not a product on the shelf.
The uncomfortable but honest answer is that aging is turning out to be both more malleable and more complicated than anyone hoped. Each breakthrough opens a door to new possibilities and to new risks we barely understand. Over the next decade, we’ll likely see targeted longevity therapies arrive first for specific diseases or organs, while broader “rejuvenation” remains experimental. In the meantime, the most powerful step any of us can take is to stay healthy enough, long enough, to benefit from whatever comes next. If real age reversal does become common in our lifetime, the people most likely to enjoy it will be the ones who did the basics well enough to still be around when it finally arrives.




