Somewhere deep in the ocean, on a quiet forest floor, and even inside our own bodies, there are organisms quietly bending one of the harshest laws of nature: everything ages, everything decays. For a long time, that was taken as an unbreakable rule. Yet in the past few decades, biologists have been uncovering creatures that seem to press pause on aging, repair themselves endlessly, or reset their life clock as if death were just an optional setting.
It sounds like science fiction, but it’s real biology, observed in labs and in the wild. I still remember the first time I read about an “immortal” jellyfish; it felt less like a news story and more like a spoiler from the future. The closer scientists look, the clearer it becomes: nature has already solved many of the problems we struggle with in human aging. The big question now isn’t whether biological immortality exists – it’s what, if anything, we can safely learn from it.
The Jellyfish That Rewinds Time: Turritopsis dohrnii

Imagine hitting a reset button on your own body, returning from adulthood back to childhood, with all your memories gone but your cells completely renewed. That’s basically what the tiny jellyfish Turritopsis dohrnii can do. When it’s stressed, injured, or starving, instead of simply dying, it transforms its mature body back into a younger, simpler stage called a polyp and starts its life cycle over again.
This isn’t just a weird party trick; it’s a full cellular reprogramming event. Cells that were specialized, like muscle or nerve-like cells, can revert to a more flexible, stem-cell-like state and then become new types of cells as the jellyfish regrows. Researchers are fascinated because this resembles some of the reprogramming methods used in cutting-edge stem cell science, except nature has been running this routine for far longer than humans have had laboratories. There’s still a lot we don’t understand, but this jellyfish shows that “one-way aging” is not a universal rule.
Hydra: The Tiny Creature That Refuses to Age

Hydra are small freshwater animals that look like living threads with tentacles, and they have become legends in aging research. In carefully controlled lab experiments, some hydra populations showed no detectable increase in death rates with time, even when observed over many years. In other words, they did not seem to age in the usual sense at all. Statistically, a very young hydra and an older hydra had roughly the same chance of surviving.
Their secret lies in a remarkable ability to constantly renew their tissues. Hydra are packed with stem cells that keep dividing and replacing older cells at a steady pace, like a house that’s being subtly rebuilt every day without ever needing a full renovation. If you cut a hydra into pieces, each piece can regenerate an entire new animal. For scientists, hydra are living proof that continuous renewal and efficient cell repair can, at least under ideal conditions, sidestep typical aging. For the rest of us, they’re a reminder that some of nature’s most radical tricks hide in the smallest, most overlooked creatures.
Planarian Flatworms: Masters of Regeneration
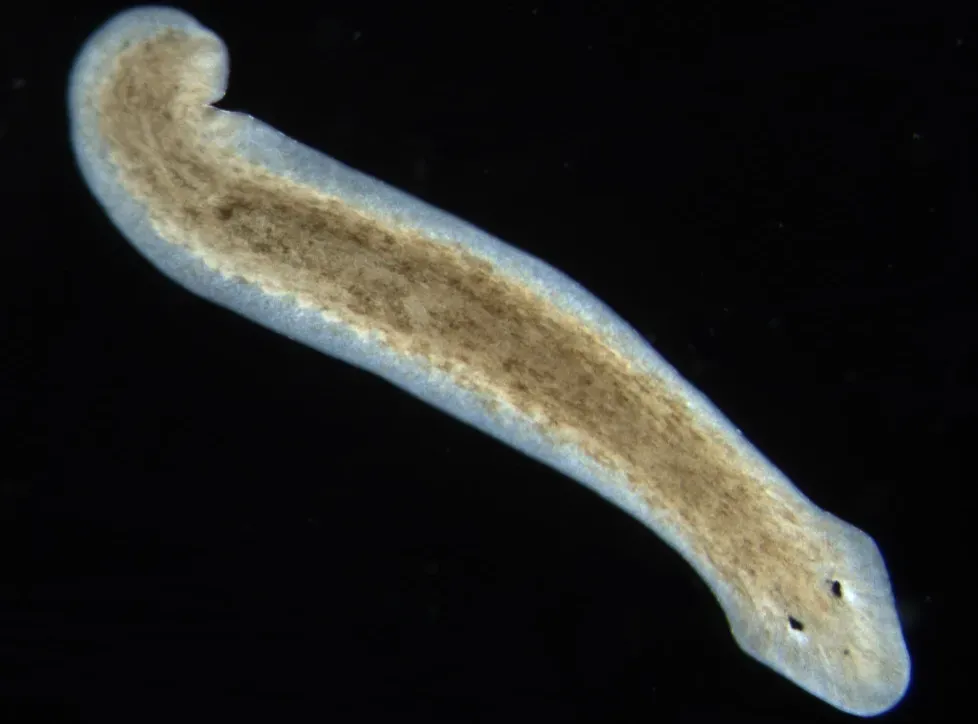
Planarian flatworms are the creatures you’d design if you wanted a biological backup system for almost everything. Slice one into multiple pieces, and each fragment can regrow into a complete worm, including a fully functional brain. They do this using a huge population of stem cells, called neoblasts, that can transform into nearly any cell type the body needs. It’s regeneration on a scale that makes lizards regrowing tails look almost modest.
What makes planarians particularly interesting for aging is how they maintain their DNA and cellular health through so much division and repair. Over time, constant cell division can lead to genomic damage, which is a major driver of aging in many species. Planarians appear to manage this with robust DNA repair systems and careful control of which cells keep dividing. Some species of planarians show extremely long-term survival and no clear aging pattern under lab conditions. It’s not immortality in the magic sense, but it’s a serious challenge to the idea that complex tissue renewal must always run down.
Lobsters and the Myth of Everlasting Youth

Lobsters have gained a bit of a reputation online as ageless, unkillable monsters of the sea, and while that’s exaggerated, the truth is still impressive. Unlike many animals, lobsters do not appear to go through a clear aging phase in the way humans or mice do. They continue to grow throughout life, stay fertile even at older sizes, and maintain strong regenerative abilities in their limbs and tissues. In the wild, the biggest limit on a lobster’s life is often environmental stress, disease, or predators, not an internal timer running out.
One key to this durability is an enzyme called telomerase, which helps maintain the protective caps on the ends of chromosomes, known as telomeres. In many animals, telomeres shorten over time, contributing to aging. Lobsters, on the other hand, keep telomerase active in many tissues across their lifespan, slowing down that erosion. Still, they are not truly immortal; molting becomes harder and more dangerous as they grow, and large lobsters can die because their own growth becomes a burden. The myth of the endlessly youthful lobster is a bit too romantic, but the biology behind it is pushing scientists to rethink how flexible aging mechanisms can be.
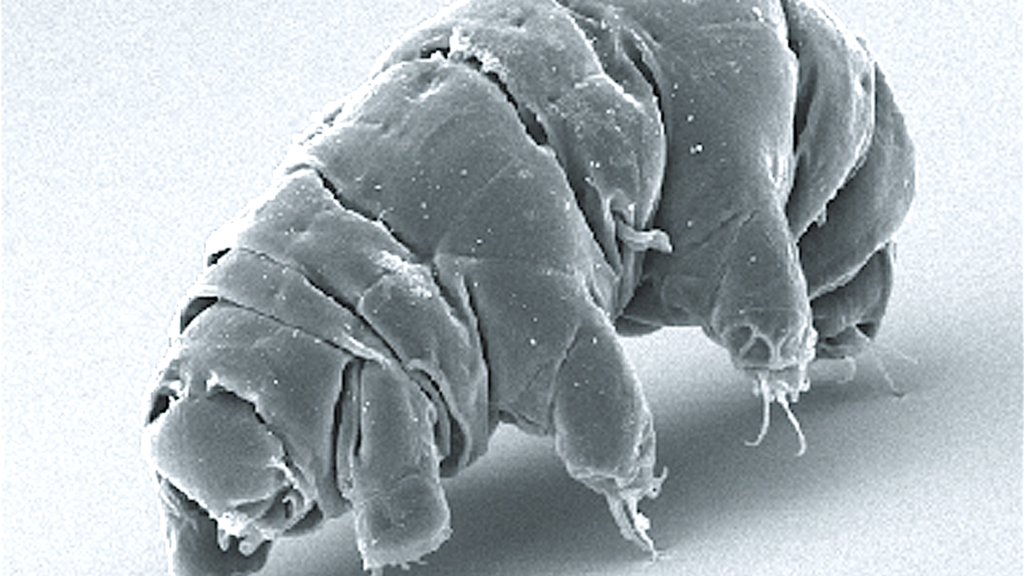
Tardigrades: Survivors, Not Immortals

Tardigrades, often called water bears, are probably the most overachieving animals when it comes to survival. They have lived through freezing, boiling, crushing pressure, radiation, and even the vacuum of space in experiments. But their form of “immortality” is different: instead of endlessly renewing themselves, they can effectively pause their biological activity to almost zero in a state called cryptobiosis. In this state, they lose almost all water, shrivel up, and can sit there for years until conditions improve.
This makes tardigrades incredible time travelers rather than classic ageless organisms. When they rehydrate, many can return to normal function, repair damage, and continue life as if they had just taken a very extreme nap. They do eventually die, and they do have an aging process when they are active, so they are not immortal in the strict sense. But their ability to suspend metabolism and protect their cells from damage has inspired research in fields like cryopreservation, dehydration-tolerant crops, and long-term storage of biological material. They show that sometimes the best way to beat decay is to slow time to a crawl.
Bacteria and the Logic of Endless Division
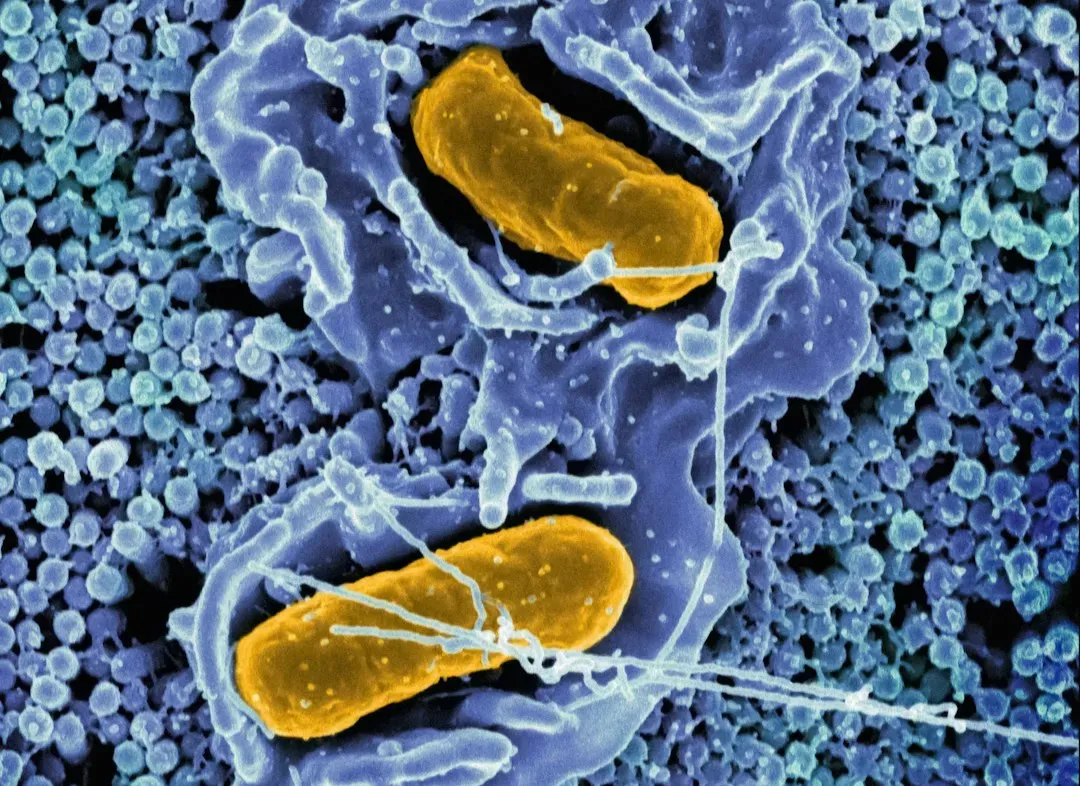
At first glance, bacteria seem like the simplest example of biological immortality. They divide, and then divide again, and their descendants can, in principle, go on forever. Each division creates two new cells, and there’s no clear “parent” that grows old and dies in the same way as a human body does. For a long time, this led to the idea that single-celled organisms might escape aging entirely. But closer studies painted a more nuanced picture: some bacteria actually show signs of asymmetry, where one side of the cell takes on more damage than the other during division.
That means some bacterial lineages carry more of the cellular “junk” as generations go on, behaving a bit like older cells, while other lineages remain comparatively “younger.” Still, as a collective, bacterial populations effectively achieve something close to immortality through continuous reproduction and adaptation. They replace parts of themselves across generations instead of relying on the same body for decades. This kind of immortality isn’t about one organism living forever; it’s about life as a pattern that never quite stops, changing shape but passing on the core blueprint again and again.
What These Organisms Teach Us About Human Aging

When you put all these stories together – the jellyfish that rewinds itself, the hydra that seems ageless, the worms that rebuild from scraps, the lobster that repairs its chromosomes, the tardigrade that pauses life, the bacteria that divide endlessly – you start to see aging not as a fixed law, but as a strategy. Different species have evolved different trade-offs between repair, reproduction, and survival in harsh environments. Humans, with our long childhoods, complex brains, and social lives, landed on a particular balance that includes aging as part of the deal.
Still, studying these “rule breakers” is changing how we think about our own biology. Researchers are exploring how to harness principles like stem cell renewal, DNA repair, controlled cellular reprogramming, and metabolic slowdown to delay age-related disease and extend healthy years of life. We’re nowhere near turning humans into immortal jellyfish, and there are serious ethical, social, and medical questions about how far we should even try to go. Yet the existence of these organisms proves that nature already knows how to soften, rewrite, or sidestep aging. The real challenge now is deciding what kind of future we actually want, now that we know immortality in some form has always been quietly swimming, crawling, and dividing all around us.