Most people never stop to think about where everything around them actually comes from. The iron in your blood, the calcium in your bones, the gold in jewelry, even the oxygen you’re breathing right now. It’s easy to take these things for granted. They just exist, right?
Here’s the thing, though. Every single one of those elements has an origin story that stretches across billions of years and involves some of the most extreme, violent, and bizarre processes in the entire cosmos. The universe isn’t just sitting still with a fixed inventory of materials. It’s actively forging new elements every single day, in ways that sound more like science fiction than reality.
Think about it. While you’re reading this, somewhere out in the vast darkness of space, stars are colliding, exploding, and transforming matter in conditions so extreme that we can barely replicate them in laboratories. The mechanisms behind element creation are stranger and more diverse than most people realize.
Stars Are Cosmic Forges That Never Stop Working

Stars spend their lives burning hydrogen into helium, then progressively fusing heavier elements like carbon, oxygen, and silicon. It’s basically a conveyor belt of nuclear fusion happening deep inside their cores. The temperatures there reach millions of degrees, hot enough to slam atomic nuclei together with enough force that they stick.
This stellar nucleosynthesis creates many of the lighter elements, up to and including iron and nickel in the most massive stars. The process releases enormous amounts of energy, which is why stars shine. Iron is the heaviest element that can be produced via fusion in a star’s core, as its fusion does not release energy but instead consumes it. After that, things get complicated.
When Stars Die, They Scatter Their Creations Across Space

Eventually, every star runs out of fuel. While a low-mass star will slowly eject its atmosphere via stellar wind, forming a planetary nebula, a higher-mass star will eject mass via a catastrophic supernova explosion. These explosions are ridiculously violent. They briefly outshine entire galaxies.
When the shockwave moves outwards through the outer layers of the exploding star, temperatures are high enough that elements heavier than iron are produced during the supernova. The blast scatters all these newly created elements into space. Supernova nucleosynthesis is largely responsible for the elements between oxygen and rubidium, which includes a lot of the stuff we consider essential.
These dispersed elements eventually mix with clouds of gas and dust, which then collapse to form new stars and planets. That’s how heavier elements end up in places like Earth. Our sun contains iron and other metals that were created in ancient supernovae billions of years ago, long before our solar system even existed.
Neutron Stars Colliding Create Gold and Platinum

Let’s be real, this is where things get absolutely wild. Researchers have shown for the first time that some of the heavier elements in the periodic table are created when pairs of neutron stars collide cataclysmically and explode. Neutron stars are the collapsed cores of massive stars, compressed to insane densities.
A neutron star can have masses over thrice that of our sun, while only being about 20 kilometers across. When two of these monsters spiral into each other, the collision is spectacular. The collision originated 130 million light years from Earth, and the cosmic merger emitted a flash of light which contained signatures of heavy metals.
The magnitude of gold produced in one merger was equivalent to several times the mass of the Earth. Think about that for a second. One collision, billions of years ago, created more gold than exists on our entire planet. The light carried the telltale signature of the neutron-star material decaying into platinum, gold and other so-called “r-process” elements.
The Rapid Neutron Capture Process Is Element Creation on Steroids

In the r-process, an atomic nucleus is bombarded with neutrons so it swells to an unstable size, but the whole thing happens so fast the element doesn’t have time to split apart. It’s like trying to inflate a balloon while it’s also trying to pop, except you’re doing it so quickly that the balloon just keeps getting bigger.
The r-process can produce the entire range of heavy elements in one spectacular flash of creation that barely lasts a second, as neutrons slam into nuclei one after another before they have time to decay. This is completely different from the slow neutron capture that happens inside certain types of stars over thousands of years.
By reanalysing data from a 2017 merger, scientists identified the signature of strontium in the kilonova fireball, proving that neutron star collisions create this element in the universe. Strontium is what gives fireworks their brilliant red color. Every time you watch fireworks, you’re literally watching atoms that were forged in the collision of dead stars.
Cosmic Rays Chip Away at Atoms to Create Rare Elements

Here’s a process most people have never even heard of. Cosmic ray spallation is a set of naturally occurring nuclear reactions causing nucleosynthesis; it refers to the formation of chemical elements from the impact of cosmic rays on an object. Cosmic rays are high-energy particles zipping through space at nearly the speed of light.
When a cosmic ray particle impacts with matter, including other cosmic rays, the result is the expulsion of particles like protons, neutrons, and alpha particles from the object hit. This process is happening right now, in Earth’s upper atmosphere and even in the rocks beneath your feet.
Cosmic ray spallation is thought to be responsible for the abundance in the universe of some light elements – lithium, beryllium, and boron – as well as the isotope helium-3. Almost all of the elements lithium, beryllium, and boron are believed to be generated by spallation, although some lithium and beryllium are thought to have been produced in the Big Bang. These elements are actually quite rare in stellar fusion, so spallation is their primary origin story.
The Big Bang Made the Universe’s First Elements

Let’s rewind to the very beginning. The first nuclei were formed a few minutes after the Big Bang through nuclear reactions in a process called Big Bang nucleosynthesis, occurring within the first three minutes of the beginning of the universe. The entire universe was a hot, dense plasma of elementary particles.
Without major changes to the Big Bang theory, BBN resulted in mass abundances of about 75% hydrogen-1, about 25% helium-4, about 0.01% of deuterium and helium-3, and trace amounts of lithium. That’s it. Around twenty minutes after the Big Bang, the temperature and density became too low for any significant fusion to occur, and the elemental abundances were nearly fixed.
Everything else we see in the periodic table had to wait. Carbon, oxygen, iron, gold, uranium – none of those existed yet. The universe was basically just hydrogen and helium for hundreds of millions of years until the first stars formed.
Black Holes Might Also Be Element Factories

Nucleosynthesis may happen in accretion disks of black holes. This is still being studied, but it’s honestly a mind-bending idea. Black holes are usually thought of as destroyers, things that swallow everything. The notion that they might also be creating elements challenges that simplistic view.
When material spirals into a black hole, it forms a superhot disk of matter. The conditions in these disks are extreme – temperatures in the millions of degrees, intense radiation, magnetic fields that would tear molecules apart. It’s possible that nuclear reactions happen there too, adding to the universe’s element production in ways we’re only beginning to understand.
Even Supernovae Have Surprising Element Creation Secrets
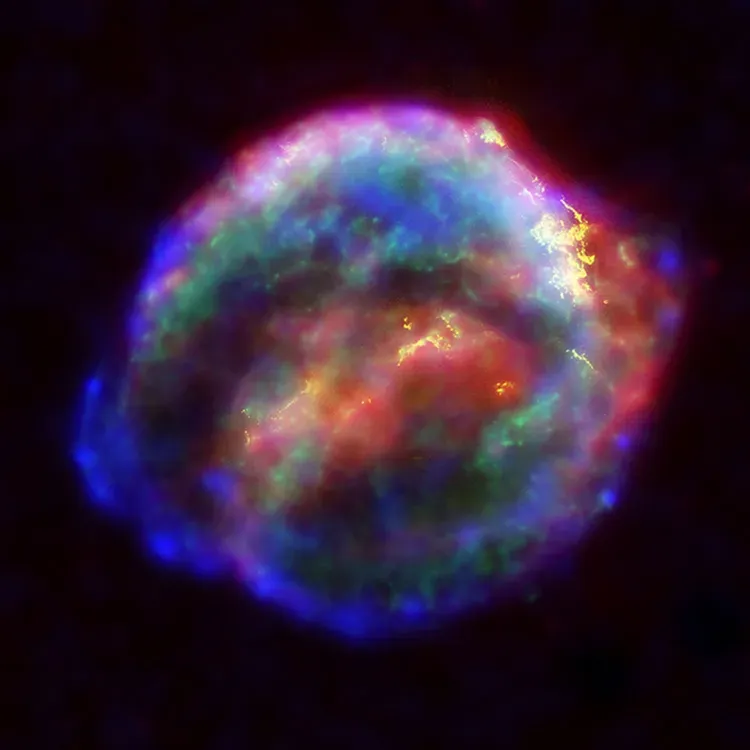
Most of the nucleosynthesis in the mass range from silicon to nickel is caused by the upper layers of the star collapsing onto the core, creating a compressional shock wave rebounding outward; the shock front briefly raises temperatures by roughly 50%, causing furious burning for about a second. This explosive nucleosynthesis happens in the last moments of a massive star’s life.
Different types of supernovae produce different element ratios. Supernova nucleosynthesis primarily occurs during Type II supernovae, caused by the gravitational collapse of massive stars, with temperatures and pressures so extreme they allow for rapid nucleosynthesis, leading to the formation of elements like gold, silver, and uranium. The exact mix depends on the star’s mass, composition, and how it explodes.
Honestly, the more we learn about supernovae, the more complicated they get. Recent simulations suggest that even the way protons interact during the explosion can dramatically change which elements get created and in what quantities.
We Are Literally Made of Stardust

We consist mostly of elements like oxygen, hydrogen, nitrogen, carbon, calcium, and phosphorus that are created through nucleosynthesis in stars that have since died. This led cosmologist Carl Sagan to famously say that we are made of “star-stuff.” It’s not just poetic language. It’s literally true.
The calcium in your bones was probably forged inside a red giant star. The iron in your blood came from a supernova. The oxygen you breathe was created by massive stars fusing helium. Every breath you take, every move you make, involves atoms that were created in cosmic furnaces scattered across the galaxy.
It’s hard to say for sure, but the atoms in your body might have come from dozens, maybe hundreds of different stellar events over billions of years. They drifted through space, collected in nebulae, condensed into our solar system, and eventually ended up in you.
The Universe Keeps Enriching Itself Over Time
Elements created through stellar nucleosynthesis are dispersed into the interstellar medium through stellar winds and supernova explosions, enriching the material from which new stars and planets can form. This is an ongoing cycle. The universe gets progressively richer in heavy elements with each generation of stars.
There is a qualitative difference between Big Bang nucleosynthesis, which was confined to the first few minutes when the entire universe was hot enough to participate, and the slow but steady contributions of stellar nucleosynthesis over billions of years. The Big Bang gave us the raw ingredients. Stars have been cooking up the complicated stuff ever since.
This chemical evolution is still happening right now. Lower-mass stars enrich the universe over a period of about 10 billion years, more than half the Universe’s present age. We live in a universe that is constantly changing its elemental composition, becoming more diverse and complex over cosmic timescales.
Conclusion

The universe’s element creation story is far stranger and more wonderful than most people realize. From the nuclear furnaces inside stars to the catastrophic collisions of dead stellar remnants, from cosmic rays chipping away at atoms to the primordial soup of the Big Bang itself, elements are being created through an astonishing variety of processes.
Every time you hold something in your hand, you’re touching matter with an origin story that spans billions of years and crosses unimaginable distances. The universe isn’t static. It’s a dynamic, evolving chemical factory that never stops working, constantly transforming simple hydrogen into the rich tapestry of elements that make up everything we see. What’s your favorite element, and have you ever wondered where it truly came from?




