We are living through one of the most extraordinary periods in medical history. Somewhere in a lab right now, a baby is thriving who would have died without a treatment that didn’t even exist two years ago. Diseases once considered death sentences are being dismantled at the genetic level. The tools scientists now have at their disposal would have seemed like pure science fiction even a decade ago.
What’s coming isn’t just incremental improvement. It’s an entire reshaping of how the human body is understood, diagnosed, and healed. From personalized cancer vaccines to gene editing that works inside your living cells, the pace of progress is staggering. Let’s dive in.
1. CRISPR Gene Editing: Rewriting the Human Blueprint
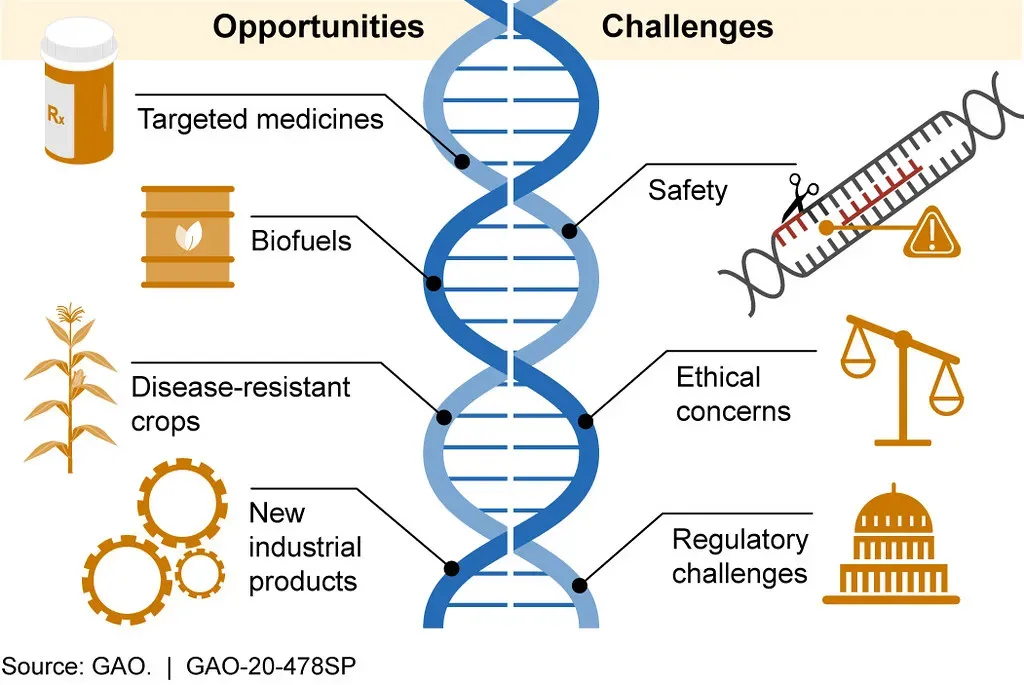
Honestly, if you had to pick just one breakthrough to watch this century, CRISPR would be it. Genome editing has evolved from a theoretical concept to a powerful and versatile set of tools, with the discovery and implementation of CRISPR-Cas9 technology propelling the field further into a new era, where this RNA-guided system allows for specific modification of target genes, offering high accuracy and efficiency. Think of it like a precision spellchecker for your DNA. Instead of just flagging an error, it actually fixes it.
In a historic medical breakthrough, a child diagnosed with a rare genetic disorder was successfully treated with a customized CRISPR gene editing therapy by a team at Children’s Hospital of Philadelphia and Penn Medicine. The infant, KJ, was born with a rare metabolic disease known as severe carbamoyl phosphate synthetase 1 deficiency, and after spending the first several months of his life in the hospital on a very restrictive diet, KJ received the first dose of his bespoke therapy between six and seven months of age. The treatment was administered safely, and he is now growing well and thriving. That is not a small thing. That is a child alive and walking because science moved faster than any disease could.
This landmark case paves the way for a future with on-demand gene-editing therapies for individuals with rare, until-now untreatable genetic diseases, and sets precedent for a regulatory pathway for rapid approval of platform therapies in the United States. The ripple effects of this one case are hard to overstate. It doesn’t just help one child. It proves the model works.
A one-time infusion of a gene-editing therapy using CRISPR-Cas9 safely reduced LDL cholesterol and triglycerides in people with lipid disorders resistant to current medications. In the trial, both LDL cholesterol and triglyceride levels were substantially reduced within two weeks after treatment and stayed at low levels for at least 60 days. This means CRISPR isn’t just for rare childhood diseases. It could one day replace the daily pills millions of people take for heart disease.
2. Personalized mRNA Cancer Vaccines: Your Tumor’s Worst Nightmare
You’ve heard of mRNA vaccines from the COVID-19 era. Here’s the thing though: that was just a warm-up. Researchers have developed an approach at the nexus of several important trends, pairing insights about the immune system’s response to cancer with advances in vaccine production spurred by the COVID pandemic, the rise of AI algorithms, and the plummeting cost of genetic sequencing. The result is something almost shockingly elegant: a vaccine built specifically around your cancer.
Once the genetic sequencing of a tumor is identified, an mRNA vaccine can be designed to target tumor-specific neoantigens, prompting a robust immune response. When compared to traditional treatments, these vaccines have been reported to have better efficacy as well as specificity. It’s like giving your immune system a wanted poster, custom-printed with the exact face of your personal cancer cells. The personalized mRNA vaccine developed by Memorial Sloan Kettering Cancer Center in collaboration with BioNTech demonstrated remarkable efficacy in pancreatic ductal adenocarcinoma patients, with vaccine-induced immune responses persisting for nearly four years after treatment in some patients. This breakthrough addresses one of the most challenging malignancies, where only roughly one in eight patients survive five years.
Today there are at least 50 active clinical trials in the U.S., Europe and Asia targeting more than 20 types of cancer. A melanoma trial led by Moderna and Merck has reached phase 3, the last step before a medicine can be approved for public consumption, and personalized melanoma vaccines could be available as early as 2028, with mRNA vaccines for other cancers to follow. It’s hard to say for sure when the floodgates will open, but the trajectory is unmistakable.
Clinical breakthroughs, particularly the success of mRNA-4157 combined with pembrolizumab in melanoma patients, have demonstrated significant improvements in efficacy, with a reduction of nearly half in recurrence risk compared to checkpoint inhibitor monotherapy. That is not a marginal improvement. That is the kind of number that changes clinical guidelines entirely.
3. Artificial Intelligence in Disease Detection: A Doctor That Never Sleeps

Let’s be real: doctors are extraordinary, but they are also human. They get tired. They miss things. AI enhances diagnostic accuracy and speeds decision-making by integrating diverse data sources, such as electronic health records, medical imaging, genomic profiles, and scientific literature. Deep learning algorithms have shown remarkable success in detecting abnormalities across various imaging modalities, including X-rays, CT scans, MRIs, and pathology slides. An AI doesn’t need coffee or sleep to catch a malignancy at 3 a.m.
Scientists using AI developed by Google DeepMind identified a previously unknown protein interaction critical to the survival of certain cancer cells. By modeling complex biological structures and interactions, the AI helped uncover molecular dependencies that are difficult to detect through conventional laboratory methods. It’s almost like having a microscope that can see around corners. Diagnostic errors affect approximately 5% of the population each year, but AI diagnostic tools are tackling this challenge through early detection and quick clinical alerts.
Scientists reported that routine eye exams could detect early signs of Alzheimer’s disease years before memory loss appears. Using high-resolution retinal imaging, researchers identified subtle structural changes and abnormal protein accumulation associated with neurodegeneration. Because the retina is an extension of the central nervous system, these changes mirror pathological processes occurring in the brain, and the method offers a non-invasive, low-cost screening tool that could be integrated into standard eye checkups.
This is especially critical for conditions where symptoms don’t become apparent until later stages, such as chronic kidney disease, where early detection can mean the difference between lifestyle changes and progression requiring dialysis. By harnessing predictive technologies, clinicians can access integrated insights drawn from blood and urine tests, medical history, and lifestyle data, all in real time, with advanced models detecting patterns that humans may miss, enabling physicians to shift from reactive treatments to proactive care.
4. Xenotransplantation: Pig Organs Saving Human Lives
I know it sounds crazy, but the future of organ transplantation might literally be pigs. The field of xenotransplantation saw several milestones, including the first genetically modified pig liver transplanted into a living human patient. The patient was a 71-year-old man with liver disease caused by hepatitis B and liver cancer who could not receive a human liver or undergo traditional surgery, and he survived for 171 days with the pig liver, showing the organ can perform essential functions in people. That’s not science fiction. That’s a real person, breathing, living with a pig liver keeping him alive.
Scientists believe that transplanting modified pig organs into people may one day help alleviate the shortage of human organ donations. In recent years, several living patients have also received pig kidneys, lungs, and hearts, with some surviving for weeks. Every one of those cases is a milestone. Each is proof that the biological barrier between species is more crossable than medicine once assumed.
The waiting list for organ transplants is an ongoing tragedy that most people don’t hear about until they’re on it themselves. Hundreds of thousands of patients worldwide wait for an organ that may never come. Xenotransplantation offers something that seemed unthinkable a generation ago: a potentially unlimited supply of donor organs, grown to match human biology through genetic modification. The ethical conversations are complex and necessary, but the medical promise is genuinely breathtaking.
5. Painless Nanoneedle Diagnostics: The Biopsy Gets a Radical Makeover

Most people dread biopsies. The needles, the recovery time, the anxiety of waiting. Now imagine getting the same information from a patch you press gently against your skin. Researchers revealed a diagnostic patch embedded with nanoneedles roughly 1,000 times thinner than a human hair, capable of collecting biological samples without causing pain. The nanoneedles penetrate only the outer layers of the skin, avoiding nerve endings while capturing proteins, genetic material, and disease biomarkers, and early studies show that the patch can extract clinically relevant information comparable to that of conventional biopsies, particularly for cancer detection and inflammatory conditions.
Because the process is painless and minimally invasive, it could enable more frequent monitoring, earlier diagnosis, and improved patient compliance. The technology may also reduce the need for anesthesia, surgical procedures, and recovery time associated with traditional tissue sampling. Think about what that means in practice. You could check for early signs of cancer the same way you check your blood pressure at home. Routine. Low-stakes. Potentially life-saving.
The painless, minimally invasive nature of this technology could enable more frequent monitoring, earlier diagnosis, and improved patient compliance. It may also reduce the need for anesthesia, surgical procedures, and recovery time associated with traditional tissue sampling. Still, more validation work is needed before this becomes a clinical standard. It is a real and exciting step forward, not a finished product. Yet given the pace of progress, “not yet” keeps becoming “now” faster than expected.
6. Epigenetic Editing and the Science of Reversible Gene Control
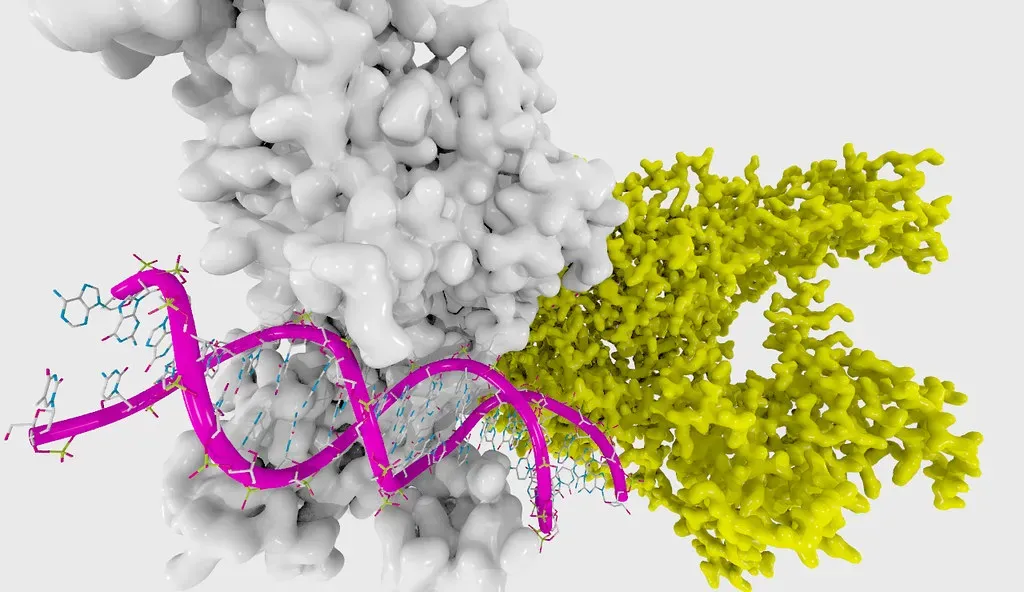
A new CRISPR breakthrough shows scientists can turn genes back on without cutting DNA, by removing chemical tags that act like molecular anchors. The work confirms these tags actively silence genes, settling a long-running scientific debate. This gentler form of gene editing could offer a safer way to treat sickle cell disease by reactivating a fetal blood gene, and researchers say it opens the door to powerful therapies with fewer unintended side effects. This is subtler than traditional CRISPR, and that subtlety might be the point.
Traditional gene editing cuts DNA. That gets the job done, but it comes with risks. Epigenetic editing is more like adjusting the volume on a gene rather than ripping out its wiring entirely. Many genetic conditions involve genes that are improperly turned on or off, and adjusting methyl groups may provide a way to correct those problems without damaging DNA. Researchers are excited about the future of epigenetic editing, noting that it allows boosting gene expression without modifying the DNA sequence, and that therapies based on this technology are likely to have a reduced risk of unintended negative effects compared to first or second generation CRISPR.
In 2026, larger clinical trials are testing how epigenetic clocks and other aging biomarkers respond to interventions. These studies are revealing whether observed changes reflect true biological improvement, guiding strategies to promote healthy aging. The implications stretch well beyond genetic disease into the very mechanism of human aging itself. It’s one of those areas where the more you look, the bigger the picture gets.
7. The Shingles Vaccine and the Surprising New Weapon Against Dementia
Nobody expected a shingles vaccine to become one of the most talked-about tools in dementia prevention. Yet here we are. A landmark real-world experiment gave the strongest evidence yet that the shingles vaccine could lower the risk of Alzheimer’s disease and other forms of dementia, finally giving concrete data to support a longtime hunch that viruses may somewhat increase dementia risk. People in Wales who received the shingles shot were roughly one-fifth less likely to develop dementia over the next seven years compared with those who did not get it. That is a massive finding hiding in plain sight.
Scientists cannot say for sure why getting vaccinated has these additional benefits. Still, they note that lingering viruses, such as Epstein-Barr, have been linked to dementia and other long-term health complications, and that revving up the immune system could boost overall protective mechanisms. It raises a fascinating and potentially transformative question: how many of the diseases we think of as purely neurological might actually be, at least in part, infectious in origin?
In a world-first, scientists in the United Kingdom used living human brain tissue to monitor the early stages of dementia. The team exposed healthy cells, collected during unrelated surgeries, to a toxic protein linked to Alzheimer’s known as amyloid. The ability to directly study how dementia progresses in the human brain could make it easier to find treatments that will work against it, given that there is currently no cure. This approach is the kind of conceptual leap that quietly reshapes an entire field of medicine.
The picture being assembled here is extraordinary. Dementia might be partially preventable through a vaccine you can get today, detectable through an eye exam years before symptoms appear, and studied in lab tissue at a level of detail never before possible. Three entirely different breakthroughs, converging on the same devastating disease.
Conclusion: The Century of Human Health

What unites all seven of these breakthroughs is a single idea: precision. Medicine is moving away from the blunt-force approach of treating bodies as roughly interchangeable, and toward treating each person’s biology as the unique, complex system it actually is. Your tumor, your genome, your epigenome, your immune system. All of it becomes data that science can now read, interpret, and increasingly, rewrite.
It would be naive to say the path ahead is smooth. Funding pressures, regulatory hurdles, and access inequalities remain real and serious obstacles. The stream of innovation can be sped up or slowed down by policy, funding, and institutional choices, and real progress, providing the fruits of that innovation to people who need them, takes a huge number of people and institutions working together. Progress isn’t automatic. It requires deliberate choices by societies, governments, and individuals to invest in and protect scientific discovery.
Still, the trajectory is undeniable. We are living through a period where incurable has become a temporary description, not a permanent sentence. A baby is walking today because scientists moved fast enough. Tumors are shrinking in response to vaccines built from a patient’s own biology. Eyes are becoming windows into the brain’s future. Which of these seven breakthroughs do you think will change your life first?




