You’ve probably heard that the gut is the body’s “second brain.” You’ve likely come across headlines urging you to eat more fermented food or avoid antibiotics. But here’s what most of those headlines don’t tell you: it’s not just a vague swarm of nameless bacteria running the show inside you. There are specific, individual microbial species, each with its own job, its own personality almost, quietly steering your metabolism, your immune system, your mood, and even your weight.
Your gut alone is one of the most densely populated microbial environments on Earth, home to trillions of microorganisms living in harmony with your body, helping with digestion and playing key roles in maintaining a balanced immune system. Most people walk through their entire lives never knowing these microscopic tenants exist. That’s a shame, because some of them are doing more for you than your most expensive supplements ever could. Others, when things go wrong, can quietly tip your health into crisis. Let’s dive in.
1. Akkermansia Muciniphila: The Gut Wall Guardian You Never Knew You Needed
Think of your gut lining as a brick wall. Akkermansia muciniphila is essentially the construction crew that keeps every single brick firmly in place. This bacterium lives in the mucus layer of your intestine and specializes in breaking down mucin, which is the protein matrix of your gut’s protective coating. Counterintuitively, that process actually stimulates your body to produce more mucus, reinforcing the barrier itself. It’s a bit like how a personal trainer breaks down your muscle fibers so they rebuild stronger.
A low abundance of Akkermansia muciniphila in your gut favors inflammatory processes, potentially leading to inflammatory disorders like irritable bowel syndrome, inflammatory bowel disease, and even colorectal cancer. Increases in its abundance can regulate metabolic functions and appear to exert protective effects against obesity, type 2 diabetes, and atherosclerosis. Because of this, it’s been considered a potential bioindicator of human cardiometabolic health. Honestly, this is one microbe you really want thriving inside you, and you can encourage it simply by eating more polyphenol-rich foods like pomegranates and cranberries.
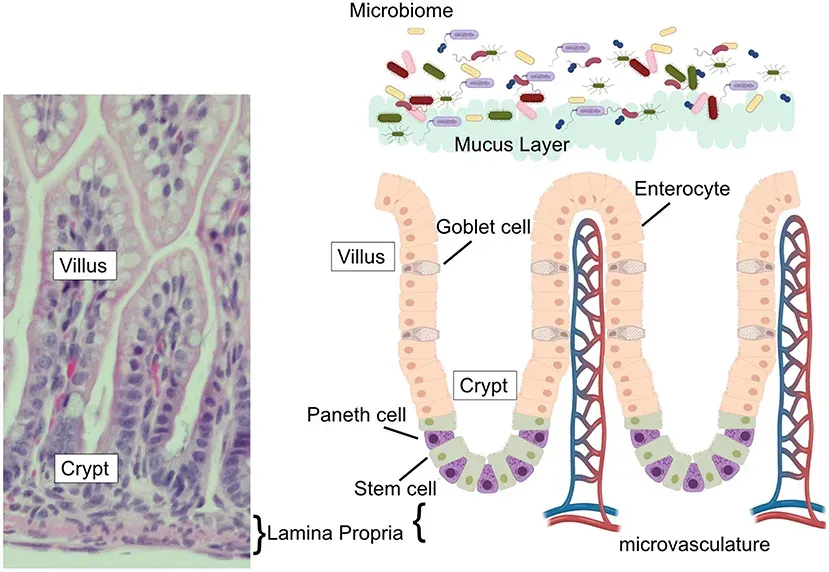
2. Faecalibacterium Prausnitzii: Your Body’s Quiet Anti-Inflammatory Powerhouse
If Akkermansia is the construction crew, Faecalibacterium prausnitzii is the fire department. It produces butyrate, a short-chain fatty acid that actively calms inflammation in your intestinal cells. In inflammatory bowel disease, patients exhibit reduced microbial diversity, with depletion of anti-inflammatory bacteria like Faecalibacterium prausnitzii and overgrowth of pro-inflammatory strains. That imbalance is not trivial. It can push your gut into a state of chronic low-grade inflammation that quietly damages tissue over months and years.
It has been established that Akkermansia and Faecalibacterium are abundant in normal populations and have protective benefits on digestive health while also enhancing the immune system, metabolism, and gut barrier of the host. What’s remarkable is that F. prausnitzii can make up roughly one in twenty of all bacteria in a healthy gut. When you lose it through antibiotic overuse, a poor diet, or chronic stress, that absence echoes across nearly every system in your body. It’s a stark reminder of just how much your lifestyle shapes who lives inside you.
3. CAG-170: The Mystery Microbe Linked to Universal Good Health
Here’s something that genuinely surprised me when I first read it. Scientists didn’t even properly document this group of bacteria until recently, yet it keeps showing up in healthy people across the globe. A global study uncovered a mysterious group of gut bacteria that shows up again and again in healthy people. Known as CAG-170, these microbes were found at lower levels in people with a range of chronic diseases, and genetic clues suggest they help digest food and support the broader gut ecosystem.
A large international study led by researchers at the University of Cambridge identified this little-known group of gut bacteria that appears far more often in healthy people. The group, called CAG-170, was consistently found at higher levels in individuals without chronic illness. The team analyzed more than 11,000 gut microbiome samples from people living primarily in Europe, North America, and Asia, and the dataset included people diagnosed with 13 different diseases, including Crohn’s disease, colorectal cancer, Parkinson’s disease, and multiple sclerosis. The fact that we’re still discovering microbes this important in 2026 tells you how much of our own biology remains a mystery.
4. Turicibacter: The Tiny Organism That May Hold the Secret to Weight Management
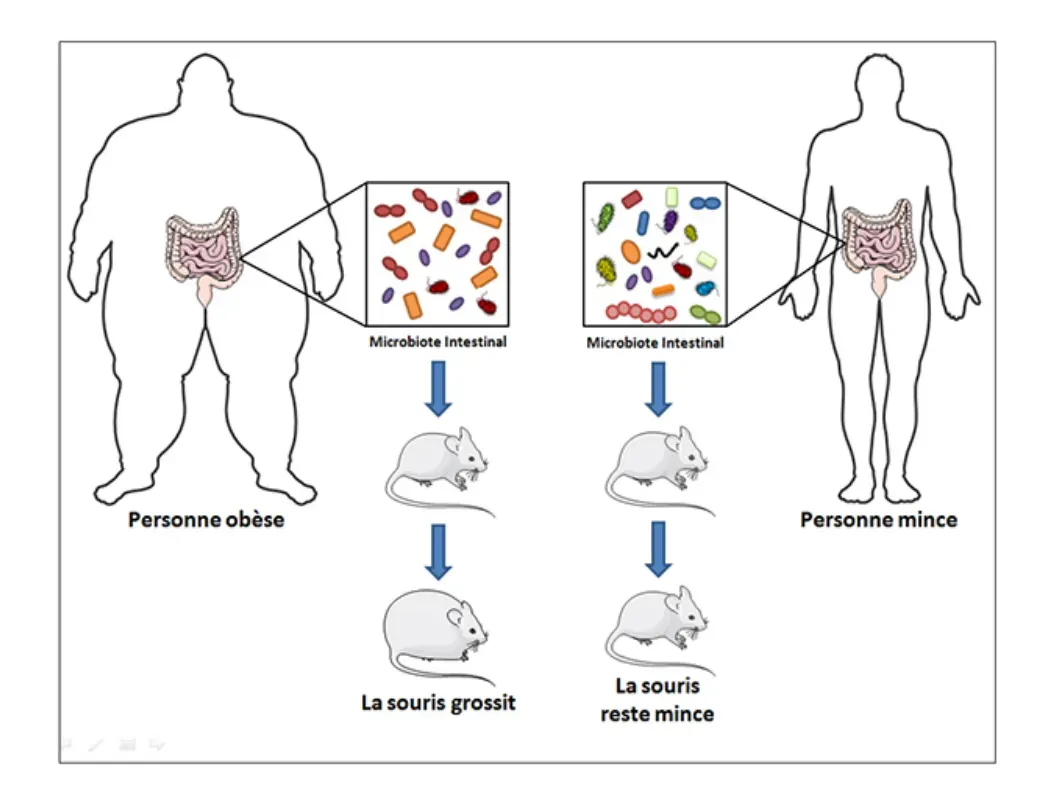
Weight management is one of the most complex, emotionally charged topics in health. You’d be surprised to learn that part of the answer might live in your gut in the form of a single little-known species. Research at the University of Utah identified a specific type of gut bacteria called Turicibacter that improves metabolic health and reduces weight gain in mice on a high-fat diet. People with obesity tend to have less Turicibacter, suggesting that the microbe may promote healthy weight in humans as well.
Turicibacter appears to promote metabolic health by producing fatty molecules that are absorbed by the small intestine, and when researchers added purified Turicibacter fats to a high-fat diet, they had the same weight-controlling effects as Turicibacter itself. The cruel irony, however, is that the bacterium won’t grow if there’s too much fat in its environment, so mice fed a high-fat diet lose Turicibacter from their gut microbiome unless their diet is regularly supplemented with the microbe. The results point to a complex feedback loop in which a fatty diet inhibits Turicibacter, and fats produced by Turicibacter improve how the host responds to dietary fats. Your diet doesn’t just feed you. It feeds, or starves, the microbes that protect you.
5. Christensenella Minuta: The Inherited Microbe That Keeps You Lean
Here’s a wild concept. What if some people are born with a genetic advantage in the form of a gut microbe? Christensenella minuta is one of the most heritable bacteria in the human microbiome, meaning your chances of having it are strongly influenced by your genes. Beneficial gut bacteria such as Akkermansia muciniphila, Adlercreutzia equolifaciens, and Christensenella minuta contribute to metabolic regulation and immune support through bioactive metabolites like short-chain fatty acids. It’s associated with lower body mass and a leaner metabolic profile in studies across multiple populations.
Recent findings suggest Dysosmobacter welbionis and Adlercreutzia equolifaciens as potential novel next-generation bacteria for addressing obesity and metabolic dysfunction-associated steatotic liver disease, respectively. Christensenella sits in this same fascinating category of next-generation microbes that scientists are eyeing for future probiotic therapies. Think of it as the microbiome equivalent of a lucky genetic hand. You can’t entirely choose who you inherit, but you can nurture what you’ve got through fiber-rich foods and a lower-sugar diet that lets these organisms flourish.
6. Oxalobacter Formigenes: The Microbe That Protects You From Kidney Stones

Kidney stones are notoriously painful, often described by those who’ve experienced them as worse than childbirth. Yet few people know that a specific gut bacterium could dramatically reduce their risk. Oxalobacter formigenes is an anaerobic bacterium with one remarkable superpower: it degrades oxalate, a compound found in many common foods like spinach, nuts, and chocolate. When oxalate isn’t broken down properly, it builds up in your urine and crystallizes into stones. It’s essentially like having a cleaning crew that prevents the pipes from clogging.
Key gut bacteria including Oxalobacter formigenes contribute to unusual health benefits such as improved gut barrier function, better glucose metabolism, reduced inflammation, weight management, and prevention of kidney stone formation. The troubling reality is that antibiotic use appears to wipe out Oxalobacter populations in the gut, sometimes permanently. The high intake of antibiotics causes an imbalance in the gut microbiota and favors systemic diseases. So the next time a doctor prescribes you antibiotics unnecessarily, there’s a real argument that your kidneys might pay for it years down the road.
7. Staphylococcus Epidermidis: The Skin Bacterium That Shields You From Infection
Most people hear “Staphylococcus” and immediately think of infections and hospital wards. But here’s the thing: not all staph is your enemy. Staphylococcus epidermidis lives on your skin almost universally, and it’s been shown to actively protect you from more dangerous pathogens. It’s the quiet security guard at the door, one you never notice until it’s gone.
When germ-free mice were exposed to Staphylococcus epidermidis, one of the most common bacteria on human skin, adding this one species of bacteria boosted immune function in the mouse skin, and the mice with S. epidermidis were able to defend against a parasite while the bacteria-free mice weren’t. Viruses and bacteria are often viewed as harmful, but researchers are drawing attention to a lesser-known side of the microbial world, highlighting the important ways microbes can support human health and challenging the idea that all microorganisms are threats. You literally carry your own biological security system on your skin every single day.
8. Clostridium Difficile: The Dangerous Opportunist Waiting for Your Guard to Drop
Not every microbe on this list is your friend. Clostridium difficile, commonly known as C. diff, is a perfect example of an opportunistic pathogen. It lives quietly in the gut of a small percentage of healthy people without causing any harm. The moment your gut microbiome is disrupted, however, usually by antibiotics, it explodes in population and releases toxins that can cause severe, life-threatening diarrhea and colitis. It’s like a dormant ember that a single gust of wind can ignite into a wildfire.
Clinical evidence demonstrates significant reductions in microbial diversity and depletion of beneficial short-chain fatty acid-producing bacteria, alongside expansion of pro-inflammatory Enterobacteriaceae and pathobionts like Clostridium difficile, particularly through the Western diet pattern. Clostridium difficile infection increases patient morbidity, mortality, and healthcare costs, and antibiotic treatment induces gut dysbiosis that is both a major risk factor for C. diff colonization and a treatment of the infection itself. That’s a brutal paradox. The medication used to treat it can also set the stage for it to return. Prevention, through a healthy, diverse diet, remains far smarter than cure.
9. Lactobacillus Species: The Familiar Name Hiding Incredible Depth
You’ve probably seen Lactobacillus on the side of a yogurt carton. It feels almost too common to be exciting. Honestly, though, that familiarity has caused most people to massively underestimate just how much these bacteria do. Different strains of Lactobacillus occupy very different roles across your body, from your gut lining to your vaginal microbiome to your oral cavity. Think of them less like a single species and more like an extended family with wildly different job descriptions.
The gut-bladder axis is also emerging as another dimension of microbiome health, and promising research suggests that probiotics may offer effective solutions to improve bacterial vaginosis and urinary tract infections that do not respond to mainstream treatments. Probiotics might help manage carbohydrate intolerance, and an integrative approach that combines adherence to a low-FODMAP diet with strain-specific probiotics may offer an effective strategy for managing symptoms associated with carbohydrate malabsorption, including FODMAP and lactose intolerance. The key word there is strain-specific. Not all Lactobacillus products are equal, and taking the wrong strain for your condition is a bit like hiring a plumber to fix your electrical wiring.
10. Bacteroides Species: The Double-Edged Sword Deep in Your Gut
Bacteroides are among the most abundant organisms in your gut, making up a significant portion of your microbial population. In healthy conditions, they’re extraordinarily useful, breaking down complex carbohydrates that your body simply cannot process on its own. Phyla Firmicutes, Bacteroides, Actinobacteria, Proteobacteria, and Verrucomicrobia contribute to the significant resident bacterial populations in the gut microbiome. They’re pillars of the entire ecosystem, the kind of organism whose presence or absence ripples through hundreds of downstream processes.
The dark side is that Bacteroides species, when they escape the gut through a perforated bowel or through immune collapse, become dangerously pathogenic. They’re a leading cause of anaerobic bacterial infections in clinical settings. The gut microbiota coevolved with humans and maintains host health by regulating metabolism, physiology, and immune functions. Different types of bacterial sensors in the gut show distinct preferences for certain classes of chemicals, which reveals that gut bacteria are not responding randomly to their environment but are selectively tuned to specific metabolic signals. Bacteroides, in many ways, exemplifies the entire story of the microbiome: context is everything. Inside the right environment, it’s your ally. Outside of it, it becomes a serious threat.
Conclusion: You Are Not Alone, and That’s a Very Good Thing
These microbes are so impactful that some researchers consider them to be a separate organ, which shapes your metabolism, your susceptibility to allergic and inflammatory diseases, and even your responses to medical treatments. That framing changes everything. You’re not just a person with a gut. You’re a walking ecosystem, and the health of that ecosystem determines far more about your life than any single medication, diet trend, or supplement ever could.
Research has identified more than 4,600 bacterial species living in the gut, and remarkably, more than 3,000 of these had never been documented there before, highlighting how much of the microbiome remains unexplored. It’s humbling, isn’t it? We’re still in the very early chapters of understanding what lives inside us. The good news is that you’re not powerless. Every fiber-rich meal, every thoughtful antibiotic decision, every fermented food you add to your plate is a vote for the microbes that are quietly fighting for you.
The microbes profiled here are not abstract science. They’re inside you right now, working in shifts, sensing their environment, producing compounds that talk directly to your cells. Some are earning their keep. Others are waiting for the opportunity to cause trouble. The question worth asking yourself is: what kind of environment are you creating for them? What do you think about the microbes living inside you? Tell us in the comments.